An ongoing dialogue on HIV/AIDS, infectious diseases,
December 8th, 2023
Clinician-to-Clinician Advice Is Great for Everyone but Still Horribly Undervalued
 One of the best things about being an ID doctor is that you get to interact with all the medical and surgical specialties. This is one of the most common answers to the question, “Why did you choose to specialize in ID?” and it certainly resonates with me.
One of the best things about being an ID doctor is that you get to interact with all the medical and surgical specialties. This is one of the most common answers to the question, “Why did you choose to specialize in ID?” and it certainly resonates with me.
As a result, we’re frequently in the position of providing clinician-to-clinician advice — which, when done without seeing the patient, was historically called a “curbside” consult. Informally asked, often prefaced with “Can I ask you a quick question,?” these curbsides have pros and cons that I’ve written about on this site numerous times.
It’s a topic near and dear to my heart, so much so that I have an entire talk on this subject that starts with several slides outlining both the benefits and the risks of this practice. And for the hard-liners (see photo above on the right) who never do clinician-to-clinician advice without seeing the patient, trust me there are benefits, some of which I’ve summarized on this slide from my talk:

In a similar theme, around 10 years ago, our hospital system started offering “eConsults,” adapting similar programs from elsewhere. Instead of referring a patient for in-person consultation to a specialist, clinicians could place an order in the medical record, and a specialist would review the question, the data from the patient’s chart, and write a note.
This form of consultation occupies an intermediate place between actually seeing, interviewing, and examining a patient, and the informal “curbside” consults where you give just general advice, undocumented anywhere. Completing the note is expected within 24 hours. Because eConsults are asynchronous, meaning we specialists don’t need to respond immediately, they are far less disruptive than getting paged or called directly.
A huge advantage of this approach over curbsides is that we can review the primary data. In addition, the consulting clinician now has specific advice documented in the chart on how to proceed, which may include ordering additional tests or modifying treatment. In a small proportion of eConsults, we advise that the clinical scenario is too complex to resolve without a formal visit. After all, we can’t put a limit on the complexity of the case — that’s at the discretion of the consulting clinician. When that happens, fortunately, we’ve still been able to weigh in on tests to order ahead of time that will make an in-person visit with us or another specialist more valuable.
How has the program done?
To say eConsults have caught on vastly understates it — they are a huge hit. Imagine if a miraculous Beatles reunion became the opening act for Taylor Swift. To quote one primary care doctor who previously had a solo practice, “It’s by far the best thing about working here.”
You can easily see why they’re so popular. Not only do clinicians get access to nearly the full range of medical and surgical expertise at our academic medical center, often an eConsult can spare their patients the inconvenience and cost of an extra doctor visit. Indeed, published data on our program strongly show that eConsults decrease the volume of formal referrals, while increasing their appropriateness. Evaluations from different centers (here and here) had similar findings. Implied in these data is that care efficiency improves and the total cost to the healthcare system declines (fewer visits = lower costs).
Sounds like these eConsults are a real advance, for patients, clinicians, and payers, a rare win-win-win trifecta — so much so that we proposed expanding the eConsult service to inpatients to deal with the never-ending deluge of curbsides that arise in hospitalized patients.
So far, all sunshine and warm tropical breezes. So, what’s the problem?
No one has figured out how to pay for these things.
In a healthcare system where procedures dominate the payment landscape — the more, the better! — an advance in efficient patient care just can’t get the attention of the payers or the administrators holding the purse strings. Yes, there are billing codes for clinician advice activities without seeing the patient, but the net pay is poor (average 0.5-1 RVU, latest conversion factor $33.06/RVU), and the technical hurdles of getting reimbursement can be tricky to implement. Plus there’s this requirement:
THESE REQUIRE INFORMED PATIENT CONSENT! Since the consulting physician is not seeing the patient, the requesting physician must obtain and document informed consent.
And guess what? If you or another ID doctor ends up having to see the patient and do a consult on the case within a week, that “formal” consult can’t be billed! As a result, hardly anyone uses these codes, at least based on my emailing a variety of ID doctors in academic medical centers, and by the lukewarm responses to an online query.
So if we’re not billing insurance, how do eConsults get supported? Note I’ve used the common euphemism for money — support.
Here, our hospital system pays us for eConsults out of the Physicians’ Organization budget, a finite resource; they set the reimbursement at $50/eConsult when we started, an amount that hasn’t budged despite a substantial increase in the cost of living over the last decade. In addition, they declined our offer to expand eConsults for inpatients, even though we offered to expedite these to make discharges more efficient.
I queried a bunch of other ID doctors in a variety of practice settings about their payment models, and their responses were diverse (and fascinating). Some get paid a set amount, like our program. One does use the “interprofessional consults code” and makes do (unhappily) with the meager payments. Another uses these codes, but augments them to what seem to me to be much more reasonable rates:
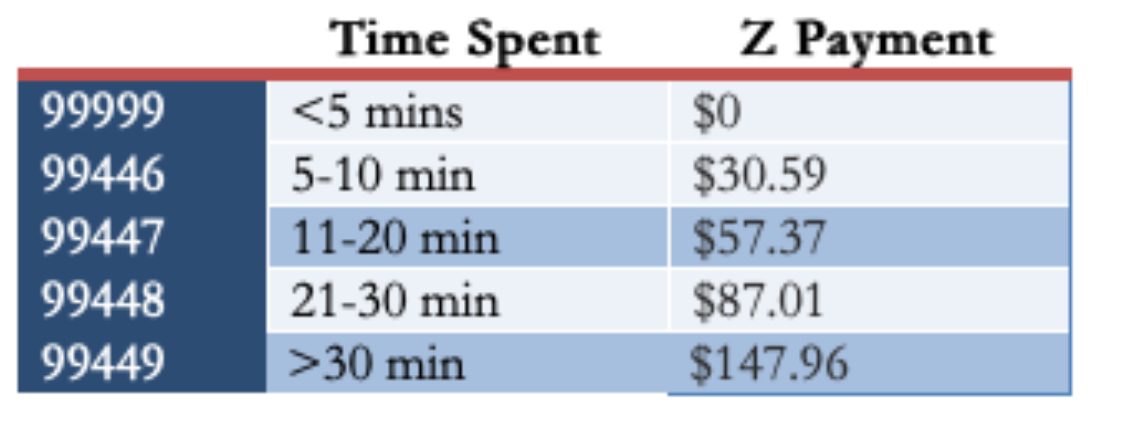
(For the record, in my experience, most eConsults would be in the third or fourth category, occasionally fifth if they involve contacting the consulting clinician or a reference laboratory.)
The ID director at a “closed” healthcare system says they have no specific payment for eConsults, but that they are tied directly to the ID doctor’s job responsibility. He estimated that “all clinician-to-clinician advice for the on-call ID doctor, without direct patient contact, accounts for 3–5 hours/day” — a substantial time commitment! Finally, Dr. Allison Nazinitzky, who does ID locums work, shared with me that one of her telehealth contracts pays her $125 for an “interprofessional consult with note — a flat fee no matter how long.”
(Check out my interview with Dr. Nazinitzky. Her experience finding the true value of ID clinical care is dynamite.)
The medicolegal aspects of eConsults should be factored into any estimation of the value of the program. eConsults may reduce medicolegal risk for the referring clinician, but they substantially increase it for the specialist taking it on. Why? As noted in an excellent review of the legal risks of curbside consults, this is because we are now explicitly reviewing the patient’s chart and providing specific advice:
The closer a physician gets to providing very specific information—what dose to start with, when to draw labs and what other kinds of studies to get, and what to do specifically with a particular patient—the closer that physician is coming to being part of the care team, as opposed to just providing general information.
Despite the boilerplate language we insert at the end of each note indicating the limitations of eConsults, this will serve as little protection if a suit is filed. As part of the care team, our names can be included in any filed suit. And even if we are later dropped because we never saw the patient, the process itself can be long and painful.
So what’s the solution? I say include these advice services as part of what constitutes the job of a clinical ID doctor — give us appropriate RVU or full-time equivalent (FTE) credit for it — and make it commensurate with the time, value, and medicolegal risk associated with the service.
And if that doesn’t happen, let’s do what Dr. Andrew Pavia recommends, in my response to the query about whether he uses the interprofessional consults codes:

In other words, stop doing eConsults until they’re properly supported. Seems like a good plan!
November 23rd, 2023
Giving Thanks — This Time to You, Readers of This Thing

Louie with his favorite dessert plates
Most years around this time I post some ID-related things to be grateful for — a research advance, a nice shift in epidemiology showing fewer people are sick and more are living longer, a new drug approval we’ve been eagerly awaiting. That kind of thing.
Last year, for example, it was gratitude that mpox had come under control (fingers crossed). That dolutegravir and bictegravir were holding up just fine. That Covid-19 finally seemed to be trending toward true endemicity (second pair of fingers crossed). And a quick comment about a certain social media site that remained a “wonderful and entertaining place to learn”, chaos at the top notwithstanding (yeesh, still holding out hope).
This year, I’m doing something different.
I’m going to thank you, whoever’s out there reading this thing, for sticking with it all these years. I’m truly grateful for your emails, your comments, your feedback, our chats at professional meetings, but most especially, for your eyes just reading this. Face it, if no one were reading these posts, I highly doubt the fine people at NEJM Journal Watch would continue supporting it. So thank you so much, and keep reading!
(Actually, if no one were reading it, I’d probably be writing something somewhere anyway — that’s how I’m wired — but you know what I mean.)
At this year’s IDWeek, at the Medical Education Community of Practice meeting, I met Dr. Priya Kodiyanplakkal, an ID doctor from New York. In the pre-meeting chat, she gave me the greatest compliment I could imagine about this blog, one she later kindly posted in this convention center lobby photo:

What she said is exactly what I’m trying to do — to convey what it’s like practicing clinical ID, the “everyday realities of being an ID physician” for all of us out there doing it.
And while I can’t know exactly who the readers are, I know from the numbers that it’s not just ID docs — it’s a much larger group of healthcare providers, people who find ID interesting, or enjoy my takes on broader issues of clinical practice (e.g., rants against fax machines, ABIM recertification, CME requirements), or just want to see the latest photo of Louie.
(My wife Carolyn purchased those dessert plates from a store which had them on sale in a remainder bin. Can you believe it?)
But whoever you are, I do know that we’ve built a remarkably nice, smart, and civil community. At one of our editor’s excellent suggestions, we have a moderated comments section, meaning we review the comments before posting. With few exceptions most of them are thoughtful and not at all nasty — it’s uncommon that we have to ding one for breaking the site’s rules. A rarity for internet communication!
Speaking of the editors, thank you also to the three of them, who take turns reviewing each post before it appears, scanning for typos, unclear prose, and unauthorized material. They’ll probably not be happy with me that this one went up without their approval, but this is a national holiday, after all — don’t want to bother them. Plus, I can guarantee that I hold all the rights to that photo at the top.
So after starting writing here in 2008, and 877 posts later, I’m still at it — thanks to all of you readers.
Hey — you can still buy these plates on eBay, in case you’re wondering. Happy Thanksgiving, everyone!
November 16th, 2023
Being a Good Doctor — Why Are the Simple Things So Hard?
The simple things that make someone not just a doctor — but a good doctor — can slip away from us when we’re too busy, or tired, or preoccupied, or hungry. That’s why it’s wise periodically to be reminded of the “soft skills” that, while individually not tricky, together make a huge difference in how patients perceive us.
“Soft skills,” by the way, is the term coined recently by Dr. Robert Oubre, a hospitalist from Louisiana who specializes in Clinical Documentation Improvement, or CDI. Periodically imparting his wisdom on social media and in his newsletter, he recently posted something entitled, “Preventing and Protecting Against Lawsuits.” Here’s what one lawyer told him was a critical part of avoiding legal trouble during patient care:
You should practice in a way that causes your patients, and everyone you work with, to like you.
Be likable! How sensible. Because beyond lawsuit protection, I’d argue these soft skill make up critical components of patient care, and legal risk-reduction is just a corollary benefit.
Here are some of those skills he cites, in bold, followed by my comments — it’s a great list. He writes from the perspective of hospital care, but a lot of them transfer over to the clinic.
Sit. In the hospital, patients are often in bed, sometimes in a chair, but hardly ever standing. Get on their level. This small physical act greatly increases their sense that you care about them and that you’re giving them time. Logistically, it’s not always easy — family might be visiting, chairs might be occupied or have stuff on them, the room might be architecturally challenging. But try to make it happen when you can.
Call. Find out who they’ve listed as their designated family member or friend to help out, and keep that person updated. We had the importance of this action driven home to us during the dark days of the pandemic, when families couldn’t visit. One time-saving strategy is to have the patient call while you’re rounding in the room or during the office visit, and put their family member on speaker phone.
Ask, “What questions do you have?” rather than “Do you have any questions?” The former invites questions, the latter subtly discourages them.
Introduce yourself. Patients meet a ton of clinicians during their hospitalization, especially if it’s a long and complex one. Coverage changes, weekends and holidays happen, specialists aplenty get called in. For many services, the era of working extended blocks of time of consecutive days is long gone. So it never hurts to remind patients who you are — and do it repeatedly, as with rare exceptions, we are terrible with names. (You lucky few out there who are good with names are fortunate indeed!) During his wife’s recent care, Dr. Oubre posted the following — that empty, confused feeling when people fail to introduce themselves:

Use the patient’s name. There’s really no excuse for not greeting the patient by their name — after all, we all know it, it’s right at the top of the medical record. So use it — “Good morning, Mr. Smith”, “Hello again, Ms. Lopez”, “Hi, Mr. Gupta, how was your night?”, etc. (Then introduce yourself — again. See above.) If the name is unfamiliar or hard to pronounce, ask them how to pronounce it, and do your best to get it right. Plus, on those occasions when a patient changes rooms, this action provides another check that you’re not about to take a history, do a physical exam, or worst of all, do a procedure on the wrong person. (Yes, it happens.)
Acknowledge others in the room — and find out who they are. I added that last bit. Both are crucial. Let’s imagine you walk in the room, and your patient (a man) is accompanied by a woman. Try this script: “Hello Mr. White, I’m Dr. Schwartz, nice to meet you.” Turn now to the woman, and say, “And who do we have with us today?” Not — “… and is this your wife?” Most of the time it will be the spouse or partner — but not always. Everyone has made the mistake of assuming it’s a spouse, only to find out it’s the patient’s friend, or sibling, or parole officer, or daughter (ugh), or mother (ugh again). If you haven’t yet made this embarrassing generational error, learn from our mistakes and don’t do it yourself.
Corollary to this point: On rare occasions, your patient doesn’t want the extra person there, but has been forced (or even bullied) into it. Give them an opportunity to be alone with you, by asking if they’d like the person to be there during the history or during the physical exam. For young adults (ages 18-25, give or take), I typically allow the parents in the room during the initial history, then ask them to leave during the physical at which time I can ask more delicate and private questions.

American Theater Poster, 1899
Mention the NAME of other doctors that are caring for them. While you may not be able to do this for all the doctors, try at least to name the primary attending physician, or in the outpatient setting, the referring clinician. And don’t just mention their name — tell them you’ve been communicating. “I heard from Dr. Li, your primary care doctor, that you had a positive TB blood test and had some questions”, or “I know from reviewing your chart that Dr. Aslam thinks you might need a change in antibiotics.” With so many people involved in a patient’s care, it’s critical to inform them that everyone is working as a team to help them get better — “Establish a team-work mentality” is another one of Dr. Oubre’s soft skills, and this strategy affirms this team work.
Smile. Listen. Affirm their experiences. One patient I saw with severe asthma told me how grateful she felt when one of our gifted pulmonologists said something like, “That must be very scary for you” when she described the sensation of having an asthma attack. Remarkably, in all the years of seeing various clinicians, no one had ever mentioned that before. Having this doctor acknowledge her fear went a long way to establishing their very successful long-term care plan.
For our new HIV diagnoses, we make sure to acknowledge the feelings they have when they find out their HIV test returned positive — something along the lines of, “It can be hard to hear that the test is positive.” Then pause, and let them speak. We can then quickly move on to reassure them that the diagnosis is no longer a death sentence, that treatment can lead to a long and normal life, and that we’ll help them adjust to this new reality of having a chronic (but treatable!) condition.
Follow up on promises. My wonderful (and still dearly missed) late colleague Paul Farmer took this to heart more than anyone I’ve met. No matter how busy or crazy his schedule, if he told a patient he’d be back to hear more of their story, or to give them boots, he’d do it. (Here’s the boots story, #2 in the list.) I’m not saying we need to buy everyone boots — but his actions stand out as a good model for what we should aim for.
Be approachable to nurses. This statement, of course, applies to all the members of the care team. There’s no room in good patient care for outdated hierarchies or authoritarian actions. The nurses, social workers, pharmacists, front desk staff, the person delivering the food tray, transport personnel — we’re all there to accomplish the same thing, which is to get our patients better. The more we acknowledge and validate the others we work with, the better we’ll do.
Remember to look at them — not just the computer. Sure, we can consult the electronic medical record during the visit — just tell them that’s what you’re doing, and even apologize if you need to go on a lengthy search. Or invite them over to do it with you! But staring exclusively at the computer screen and typing while seeing a patient is a surefire way to create distance between “us” and “them.” And yes, I added this one to the list, as it’s long been a pet peeve of mine.
A non-medical person reading the above might think this stuff is easy. What’s the big deal? But trust me, the fact they seem so obvious stands in sharp contrast to just how difficult they are to do consistently, in particular when external demands weigh in as distractions.
But try we must — not only to avoid lawsuits (the original purpose of Dr. Oubre’s excellent list), but just to be a kind and caring doctor.
Anything left out you want to mention?
It certainly never hurts to ask about pets — people love talking about their pets, especially if they have a special talent.
October 31st, 2023
HIV Research Highlights from IDWeek 2023
 Having already featured an important non-HIV clinical research study from IDWeek — the amazing ACORN trial — I turn now to a grab bag of HIV-related studies, a veritable Halloween treat bag full of them.
Having already featured an important non-HIV clinical research study from IDWeek — the amazing ACORN trial — I turn now to a grab bag of HIV-related studies, a veritable Halloween treat bag full of them.
(Note to self: What’s with that “Halloween treat bag” reference? Couldn’t you come up with a less awkward way to link this content with the posting date?)
Note acknowledged. I’ll think of something snappier for next year!
Here are the studies:
Compared to placebo, semaglutide markedly reduced weight and total body fat in PWH with lipohypertrophy (#1984). Interestingly, there was no effect on ectopic fat in the liver and pericardium, locations which correlate with some adverse outcomes. Lead investigator Grace McComsey (who has led several remarkable single-center studies on metabolic issues) cautioned that lipoatrophy might worsen with this treatment, potentially a concern in particular for our older patients who received thymidine analogue drugs (ZDV, d4T) in the distant past.
In a large (n=225) retrospective cohort study, GLP-1 receptor agonists were more likely to lead to > 5% weight loss in those with higher baseline BMI and longer duration of treatment (#1983). Dulaglutide appeared less likely to achieve this weight loss than semaglutide and terzepatide. What does it say about the current state of outpatient HIV management that these two studies of GLP-1 agonists were featured oral abstracts at an ID meeting?
One dose of penicillin is as good as three for early syphilis (#2889). During my ID fellowship, a local expert strongly opined that 3 doses were necessary, especially with concomitant HIV. Not so say the results from this multicenter randomized trial, where response rates were comparable with 1 vs 3 doses, including in the 67% of study participants who also had HIV. Data confirm what is recommended in treatment guidelines.
A bunch of studies looked at various comparisons between BIC/FTC/TAF and DTG/3TC, two highly popular and effective regimens. In an interim analysis of a randomized clinical trial comparing remaining on BIC/FTC/TAF vs. switching to DTG/3TC, the switch strategy was non-inferior at week 24 (#1603). There were more discontinuations in the DTG/3TC arm, explained in part by the open-label nature of the study (ascertainment bias). As with the TANGO and SALSA switch studies, switching to DTG/3TC did not result in weight loss. Interestingly, a person who remained on BIC/FTC/TAF experienced virologic failure at week 12, with detection of M184V and G140S mutations. I believe this is the first report from a prospective study of any participant experiencing virologic failure with INSTI resistance on this regimen — wonder if there was prior treatment failure on RAL or EVG.
Switching to BIC/FTC/TAF (n=3527) or DTG/3TC (n= 2213) in clinical practice maintained virologic suppression, with very rare instances of virologic failure (#1023). Only two and three cases of virologic failure occurred, respectively, after over a year of follow-up. Since this is a non-randomized study, the patient characteristics differed between those getting BIC/FTC/TAF vs. DTG/3TC: BIC/FTC/TAF recipients were significantly more likely to be Black, on government insurance, have experienced prior virologic failure, and have a lower CD4 count; those on DTG/3TC had a lower eGFR. Regimen discontinuation occurred statistically earlier with DTG/3TC versus BIC/FTC/TAF.
Related to the above, clinicians gave different reasons for switching to BIC/FTC/TAF vs. DTG/3TC (#1570). For BIC/FTC/TAF, these reasons were inconsistent healthcare visit attendance, adherence challenges, substance use, HBV coinfection, or viremic on current regimens. For DTG/3TC, concerns about low eGFR or excess weight gain motivated the switch. For the weight issue, note that none of the switch studies of DTG/3TC have shown any difference between BIC/FTC/TAF and DTG/3TC when it comes to weight — and IDWeek provided another one (see #1603 above).
Out of 1843 PWH who started CAB/RPV in a US-based cohort, 229 (12%) had a detectable viral load at treatment onset, a strategy outside of licensed indications (#1028). For 93 of them, the viral load was > 200, so true virologic failure. Virologic suppression was achieved in 82% overall, with 4% experiencing virologic failure. There was scant data on pre- or post-treatment resistance, or more importantly why the clinicians chose this regimen — especially since the median CD4-cell count was around 500! As noted previously, I strongly believe that if we’re using this treatment for virologic failure, we should limit it to those with low CD4-cell counts at very high risk for disease progression.
Data from a claims database suggests that adherence and persistence on injectable CAB/RPV is better than oral ART (#1024). Adherence was generally high for both strategies, but significantly higher with CAB/RPV using the 90% adherence threshold. Data are both reassuring and perhaps not surprising, given that CAB/RPV requires fewer doses, our generally careful selection of candidates for this strategy, and the intense efforts to keep those who are on it engaged.
In a pilot study, 33 patients successfully received CAB/RPV at home (#1027). Remarkably, a single nurse drove to homes in a 3000 square mile region. The study sent the medications by mail, with instructions for home refrigerator storage and pre-visit preparation by the patients. This is a good proof-of-concept study, but for obvious reasons not sustainable by most healthcare systems. And while I once thought that pharmacies would be a good location for getting CAB/RPV, the commercial pharmacies in our region are now massively understaffed and overwhelmed.
In a prospective cohort study, women with low-level viremia had higher rates of treatment failure, with a trend toward more multi-morbidity (#1025). These results are similar to those observed in cohorts made up of mostly men. Management strategies are still uncertain, aside from redoubling efforts at ensuring adherence and using high resistance barrier regimens.
In large sample of MSM PWH engaged in care at academic HIV clinics with a program for anal cancer screening, only half ever received any screening (#1478). Follow-up was notably low as well. Test performance of the screening anal pap was poor — there were high rates of “unsatisfactory” results, and low rates of detection of high-grade lesions. In other words, the anal pap falls far short of required sensitivity and specificity to be a good cancer screening tool. Controversial opinion — we should stop doing anal pap smears until we have a better screening test, and in the meantime figure out which MSM with HIV should be referred directly to high-resolution anoscopy.
Hepatitis B reactivation in PWH who have isolated hepatitis B core antibody and switched to non-hepatitis B-active ART is uncommon, but does occur (#1026). These regimens lack any of tenofovir, lamivudine, or emtricitabine. Data came from a large VA cohort study, in which 5,954 were deemed at risk, and 89 (1.5%) experienced reactivation (turning surface antigen positive or having detectable HBV DNA). It’s less likely if surface antibody is also present, and much more likely if they were historically surface antigen positive (20% risk). Studies such as this one become much more clinically relevant in the CAB/RPV era.
In a cohort of PWH at an academic medical center, 23% of those with cirrhosis died during a 3-year follow-up period (#1983). This was 4 times higher than the overall HIV cohort mortality and occurred even though 90% had HIV viral suppression. Although HCV was the most common cause of cirrhosis, the study did not include details on HCV treatment history.
We’ve got PrEP, and PEP, why not PIP (“PEP in Pocket”, #2894)? In this study, 112 people at risk for HIV but with infrequent exposures were provided with 28 days of DTG plus TDF/FTC in case such an exposure occurred. Strategy allows for self-administered prevention and follow-up, a choice made by 35 of the participants, with high rates of follow-up. Some transitioning to PrEP.
That’s a wrap! Am sure I missed something interesting, as IDWeek now has a good portfolio of research studies, in addition to the great lectures, debates, and overall schmoozing.
Speaking of research, I submit the following video of a really fun research job.
October 17th, 2023
A Brilliant Strategy for Conducting Clinical Trials — The ACORN Study

Prompt: Make a cartoon battle between cefepime and pip-tazo.
The secret to doing a great clinical trial is quite simple. Here, I’ll share it with you:
- Come up with an important clinical question for which there’s true equipoise.
- Choose primary and secondary endpoints that people care about.
- Make the inclusion and exclusion criteria easy to understand and chosen so that they define a readily available and broadly applicable study population.
- Create a system in which referrals, enrollment, and informed consent are as frictionless as possible.
- Start the study, and collect the baseline and follow-up data.
- Analyze and report the results.
There, piece of cake! Off you go, time to get started.
I hope your sarcasm meter is turned on because pulling off any of the above is in fact exceedingly difficult. That’s why the ACORN study comparing cefepime with piperacillin-tazobactam (pip-tazo) — presented last week at IDWeek, and simultaneously published in JAMA — is so wonderful. And it’s not just because of its clever name: Effect of Antibiotic Choice On ReNal Outcomes.
Let’s go through the above recipe and see how they did.
Come up with an important clinical question for which there’s true equipoise. In adults hospitalized with acute infection, which antibiotic is better: cefepime or pip-tazo? Members of Team Cefepime would cite pip-tazo’s notorious association with kidney injury, especially when combined with vancomycin, and the broad anaerobe coverage, which may further disturb the microbiome.
Team Pip-Tazo would counter by reminding Team Cefepime about their drug’s neurotoxicity and say that pip-tazo’s nephrotoxicity may be artifactual — the result of inhibiting tubular secretion of creatinine, with no change in actual glomerular filtration rate (GFR) based on cystatin C measurements. Plus, Team Pip-Tazo will defend that extra anaerobic (and enterococcal) coverage as helpful for empiric therapy.
(Brief shout-out to Team Neither, made up of antibiotic stewards arguing that empiric coverage of Pseudomonas is not needed for all acutely ill people. You’re welcome.)
Choose primary and secondary endpoints that people care about. From the paper’s quick summary:
Does the choice between cefepime and piperacillin-tazobactam affect the risks of acute kidney injury or neurological dysfunction in adults hospitalized with acute infection?
That’s pretty clear. After all, the broad-spectrum coverage of these two drugs is similar, so adverse events are likely to dictate the best choice. If the study finds differences in efficacy, that would be quite interesting as secondary or exploratory endpoints. But common sense dictates that they’re probably not that different in efficacy, so setting the study up as a superiority trial on efficacy (“powering” for an endpoint of days of ICU care or death) would require a sample size so large as to make the study impractical.
Make the inclusion and exclusion criteria easy to understand, and chosen so that they define a readily available and broadly applicable study population. How about anyone in the emergency room or medical ICU for whom a clinician plans to start one of these two antibiotics? That’s pretty simple — and it must happen a zillion times a day in hospitals all over the world.
Create a system in which referrals, enrollment, and informed consent are as frictionless as possible. Here’s the true genius of the ACORN trial. After the doctor ordered either cefepime or pip-tazo in their medical center, the investigators leveraged the electronic medical record to screen automatically for critical exclusion criteria. With none found, their order triggered a prompt for the doctor to consider enrolling the patient in the trial.
During primary investigator Dr. Edward Qian’s excellent presentation at IDWeek, his slide with the image of the prompt (available on the study’s supplemental appendix) elicited gasps of amazement — at least it did from clinical trials geeks like me. This is just a brilliant way to encourage referral and enrollment. Frictionless clinical research at its best!
(Brief aside: I’m not sure about your hospital, but where I work, changes to EPIC, our electronic medical record, move forward about as fast as that slug you found under your garden’s flowerpot. What was this wizardry Dr. Qian and colleagues did to scoot this slug along?)
But what about informed consent? As discussed in detail on page 10 of the study’s appendix, it turns out informed consent to a study can be waived if a study is deemed minimal risk and the intervention is an emergency. Empiric cefepime or pip-tazo for acutely ill, hospitalized febrile adults certainly meets the criteria of standard of care (minimal risk), and the sooner antibiotics start the better (emergency). Check and check!
Start the study, and collect the baseline and follow-up data. From November 10, 2021 to October 7, 2022, 2634 participants enrolled in the study at a single center. (I told you this happens a zillion times a day — full enrollment in less than a year from just one hospital!) Patients enrolled were well-matched at baseline, with sepsis the initial diagnosis in just over half. The median duration of the initially assigned antibiotic was 3 days, but a substantial fraction received treatment for longer.
Clinicians added vancomycin in 83% of the cefepime group and 81% of the piperacillin-tazobactam group for a median of 2 days. Roughly equal proportions of both study arms had treatment modified via other antibiotic changes, including switching to the other drug (cefepime to pip-tazo in 19%, pip-tazo to cefepime in 17%), or broadening treatment by adding an aminoglycoside or switching to a carbapenem. In other words, the very common practice of antibiotic switches for critically ill patients — don’t just stand there, start meropenem! — was equally common in both arms.
Analyze and report the final results. For the primary endpoint, the highest stage of acute kidney injury or death by day 14 did not significantly differ between the cefepime group and the pip-tazo group. Even the addition of vancomycin didn’t have a differential effect. However, for the important secondary endpoint of delirium or coma, here the pip-tazo group had significantly fewer hospital days with this complication. Deaths were similar between arms, as were a bunch of other clinical and laboratory endpoints.
So Team Pip-Tazo wins, though it’s a really close call. And may the pip-tazo/vancomycin nephrotoxicity RIP, at least based on this short-term exposure.
Now no study is perfect, and the authors responsibly address the limitations in their discussion. For me, the biggest one is that as an open-label study, clinicians may have been more prone to make the diagnosis of encephalopathy with cefepime, and this turned out to be the only significant difference between the two strategies. Read the full paper and the accompanying editorial to see further discussion.
Limitations notwithstanding, the study remains an impressive feat of generating clinically relevant evidence on an important topic, and doing so quickly in a way that harmonizes with actual patient care. The novel strategy of enrolling eligible participants through the electronic medical record in such a seamless way is a model for other clinical questions in both ID and beyond.
What should we study next?
Because it can’t be as hard as doing these things on a treadmill …
October 8th, 2023
An October ID (and Non-ID) Link-o-Rama

The Lyme Laser.
For those venturing next week to IDWeek here in Boston, fall gives us our very best weather. Comfortable sunny days with brilliant blue skies, cool evenings, low humidity — great weather for exercising and sleeping. Usually you just need a light jacket. And now, after one of the rainiest Septembers on record (boo!), October has so far has been heavenly (yay!).
But you don’t come to this site for a New England weather report, which might change day-by-day anyway.
Let’s go to the links, starting with ID topics:
- Lyme vaccine clinical trials continue, and one candidate is quite far along. Fingers crossed, at least one of these two vaccines will be safe and effective, then widely adopted in highly endemic areas. My non-medical friends here frequently wonder why there is no vaccine for humans if their dogs can get one. I tell them there was one — but it got caught up in the controversial cloud that follows this disease around like dust over Pigpen.
- How’s this for an example of this dust? Or crap, to be more accurate: You can get treatment with a “Lyme Laser”. It “addresses the concern of the biofilm established in the advanced stages of Lyme disease,” and “enhances the body’s natural abilities to improve function and regeneration.” Seriously, the unvalidated and potentially dangerous “treatments” offered for this serious infection continue to strain imagination. Note that pricing on this site (and others just like it) is conspicuously absent.
- The NIH-sponsored clinical trial ACTIV6 for COVID-19 is still open, evaluating metformin. The study will (I hope) confirm the benefits seen in the COVID-OUT trial, in which metformin reduced the incidence of long COVID by 41%. Importantly, people can also take other COVID-19 treatments and still participate. I’m particularly interested to see if it can reduce the incidence of rebound after nirmatrelvir/r, which remains a bedeviling problem.
- PEPFAR may be in trouble. Could a rare example of bipartisan political support for a hugely successful public health program come to an end? That’s the worrisome message expressed in recent coverage of PEPFAR, which pays for HIV treatment in resource-constrained countries with high HIV prevalence, especially in Africa. At last estimates, the program has saved 25 million lives. Wow.
- The CDC called for comments on the use of doxycycline for post-exposure prophylaxis for STI prevention. You will not find them by clicking this link, and getting to them is tricky, so here are “guidelines” on how to find the draft guidelines: Go this YouTube video, watch the summary of the evidence, and eventually (at 15:15) you’ll reach this:

- In related news, this modeling study shows that adopting doxycycline for STI PEP using these criteria would be an efficient use of resources. Monitoring for STI resistance will be critical.
- Can oral swabs run on a multiplex PCR platform help diagnose community acquired pneumonia? In this study, correlation between oral and lower respiratory results was high when S. pneumoniae and H. influenzae were the cause of the pneumonia. Sure would be easier than trying to get an induced sputum.
- Ceftobiprole was noninferior to daptomycin in treatment of Staph aureus bacteremia. Wow, talk about a blast from the past — I thought this cephalosporin (with activity against MRSA) had long been abandoned, but here it is again! Look for this paper in many ID and medicine journal clubs over the next few months, as there are numerous questions about the study design and the clinical implications. I worry in particular about the control arm — daptomycin (not oxacillin or cefazolin) for MSSA (76% of study participants)? And was daptomycin underdosed?
- In other MSSA bacteremia research, here’s a different prospective randomized clinical trial: The study compared cloxacillin alone vs. cloxacillin plus fosfomycin. The combination regimen failed to show superiority, despite more rapid clearance of bacteremia at day 3 — reminds me of adjunctive gentamicin in this regard. Great to see these studies of Staph aureus bacteremia, which is almost certainly the most common reason for inpatient ID consults, at least in the United States.
- Herpes simplex encephalitis has a high rate of subsequent neurologic complications, with immune-mediated choreoathetosis particularly common. This in-depth study suggests corticosteroid treatment of this late complication is warranted. And it’s a reminder that despite being “treatable” with high dose intravenous acyclovir, HSV encephalitis remains a devastating diagnosis — patients and their families should be counseled up front about the potentially serious outcomes.
- The viral dynamics of COVID-19 have dramatically changed in relation to symptom onset since the start of the pandemic. Viral loads of SARS-CoV-2 now peak at day 4 of symptoms (not at or right before symptoms start), with rapid antigen testing gradually increasing in sensitivity over symptom-days 1-4. The results clearly emphasize the need to re-test in patients with negative tests early in the illness, and also have infection control implications. Our broad pre-existing immunity is responsible for this “new normal”.
- The WHO endorsed use of a highly effective malaria vaccine, the second vaccine now available. Work on malaria vaccines has been a struggle for decades, so this progress is a welcome advance indeed — because in separate news, the mosquitoes appear to be winning. Global malaria deaths are ticking up, and other mosquito-borne diseases such as dengue and chikungunya continue to spread.
- These authors provide an interesting perspective on those annoying low-level viral load results that create all kinds of anxiety — namely, that the labs and clinicians should focus on the 200 copies/mL threshold as the important one for “U=U,” as this was the lowest threshold used in the large studies evaluating HIV viral load and transmission. While I agree that this is a reasonable approach (and the one I use in clinic), there is some uncertainty around this threshold, since most of the people in these studies of transmission on ART had undetectable viral loads — the 50-200 sample was necessarily small. Nonetheless, I 100% agree that flagging these results with a big red exclamation point is unnecessary. And check out panel 4 in the figure.
- The Nobel Prize in Medicine went to Katalin Karikó and Drew Weissman, whose work led to the development of mRNA vaccines against COVID-19. Lots of chatter about how Karikó “failed” in traditional academic research, which prompted her to leave her university lab setting because she was unable to get grant support. Let’s face it, the weeding out process in academic medicine specifically and science in general will definitely overlook some innovative geniuses, as grant-review committees are famously conservative in favor of established researchers and familiar strategies.
- You know that strange sensation of time distortion since the start of the COVID-19 pandemic? “Pandemic skip” is a great descriptive term. And though the writer of this piece is just 30, it applies to every age, doesn’t it? And sometimes it feels like just a few months since those terrible dark days in 2020, and at other times it feels like it must have been many, MANY years ago, certainly much more than just 3. Don’t you agree?
- Here’s a poetic rumination on microbial odors. Impressive alliteration on display: “From the fungal fragrances that sing of environmental mold to the bacterial bouquet that warns us off week-old leftovers, our air comprises a microbial miasma of information, ripe for research and exploration.” I’m sure Cliff, the C. diff-sniffing dog, would agree!
Now, a brief non-ID section:
- Over in The New England Journal of Medicine (the Mother Ship of this Journal Watch enterprise), the editors kindly published how I felt when the news broke about a business decision that will dramatically reshape the clinical, research, and training experience in Boston — the departure of Dana-Farber Cancer Institute from Brigham and Women’s Hospital, where I work. Still can’t believe this is happening. Makes me very sad.
- These two oncologists provide a highly critical viewpoint on Maintenance of Certification by the American Board of Internal Medicine. Interesting to contrast their approach with my more ruminative account, but both ultimately come to a similar conclusion, which is that there must be something better.
- Here’s a wonderful essay for those of us of a certain age, contrasting the experience of travel both with, and without, illness. On the topic of the ailments that start accumulating as you age: “It’s as if we were all devices made by some big tech company, designed to start falling apart the instant the warranty expires and to be ingeniously difficult to repair, with zero support for older models.” Wow, wish I’d written that sentence.
- You can now order medical care on Amazon. My wife, a primary care pediatrician, says that everyone knows this by now, but somehow I missed it. But look — plenty of infections on the list! UTI, HSV, vaginal yeast infections, sinusitis. Text- or video-visit based care. And if your diagnosis is COVID-19, it’s $35 for text care, $75 for a video. Stuff like this makes you wonder about this future of healthcare, for better and (mostly) for worse, doesn’t it?
- You don’t have to be a baseball fan to find this documentary of Yogi Berra joyful and fun. Berra seemed to be the kind of person everyone loved — such a desirable trait! That he happened to be a world-class athlete (and kind of unusual looking, with quotable quotes) added to the charm. I’m sure that his very charismatic granddaughter, Lindsay, will no doubt wonder what she and her famous grandfather are doing on an ID blog.
Since people will be coming from all over the world to IDWeek, how about this impressive and nimble take on country-specific accents? Wow, what talent. What’s your favorite?
Hope to see many of you at IDWeek!
September 22nd, 2023
Long-Acting Cabotegravir-Rilpivirine for People Not Taking Oral Therapy — Time to Modify Treatment Guidelines?

Cephalopods, by Jean Baptiste Vérany.
HIV treatment guidelines are understandably reluctant to endorse practices that have limited data. Having served on two such panels (previously, DHHS and currently, the IAS-USA guidelines), I totally get this — you don’t want to put a stamp of approval on strategies that may ultimately do more harm than good.
With the caveat that I cannot speak for the guidelines panel I currently serve on, I can give you my opinion on a topic much discussed among us ID and HIV specialists — namely, the use of long-acting cabotegravir and rilpivirine in people not taking standard oral ART, despite all our efforts to encourage them to do so.
Here it is:
If the alternative is untreated and progressive HIV disease, and the patient has advanced immunosuppression, we should stop discouraging use of this potentially life-saving therapy.
I base this on what are now a few published studies, including the original reports from UCSF, a follow-up from them with a larger sample size, a modeling analysis demonstrating substantial projected survival benefits, and now — just published in Clinical Infectious Diseases — a small case series from a different clinic with comparably excellent outcomes.
At a Ryan White-funded HIV clinic at the University of Mississippi Medical Center in Jackson, clinicians began offering injectable CAB/RPV in February 2022 as a salvage option to people with HIV (PWH) who had viremia despite intensive case management strategies. Since that time, they have offered it to 12 patients — 7 women, 5 men. The baseline mean CD4-cell count was 233, and HIV viral load 152,657 copies/mL, with 5 of 12 having a history of AIDS-defining opportunistic infections.
With the caveat that follow-up is necessarily short, the results were excellent: All 12 achieved viral suppression within 3 months of treatment initiation, with no virologic rebound to date. They also reported good immunologic responses and high adherence to scheduled injections. These favorable results are all the more remarkable since some of the participants had NNRTI and INSTI resistance mutations, though none had full predicted resistance to either drug.
The experience with these dozen patients is now being reproduced anecdotally in small case numbers from other centers, including our own. While San Francisco led the way, this is doable in any HIV treatment clinic with intensive case management services.
Meanwhile, the DHHS HIV treatment guidelines continue to state the following:
The long-acting ARV combination of injectable cabotegravir (CAB) and RPV is not currently recommended for people with virologic failure [DHHS].
I strongly believe the time has come for us to revise these prohibitory statements (a similar one appears in the IAS-USA guidelines). Such statements may make it difficult for clinicians to procure this treatment for PWH whose treatment options (oral ART) otherwise just aren’t working. One can craft language with appropriate cautionary statements describing the unusual settings in which CAB-RPV is the best — and, importantly, the only — option for people with virologic failure.
Here’s a go at this:
Under rare circumstances, long-acting CAB-RPV can be an option for people with virologic failure when no other treatment options are available or effective. Such treatment must be accompanied by intensive case management and close follow-up. It should be limited to PWH who meet the following criteria:
- Unable to or cannot take oral ART
- Are at high risk of HIV disease progression (CD4 < 200 or a prior history of AIDS-defining complications)
- Have virus susceptible to both CAB and RPV
- Can be supported by intensive case management services
Sure, there will be cases where treatment failure with resistance occurs; where patients are lost to follow-up; where we will need to battle with payers to convince them to cover this non-FDA–approved indication; where the team of people (and it’s always a team) charged with case-management and medication administration spend enormous time and energy to no avail.
I acknowledge these issues and frustrations, but at the same time, ask — what is the alternative?
It can’t be to withhold the only regimen that might help them. Because as shown in the figure posted below, the median life expectancy for someone with AIDS in the pre-ART era — advanced HIV disease, no treatment — was less than 2 years.
When I was a medical resident and ID fellow, the median survival for a person diagnosed with an AIDS-related opportunistic infection was around 18 months pic.twitter.com/yXe37iDFgS
— Paul Sax (@PaulSaxMD) August 27, 2023
I think we’d all agree that this grim prognosis is simply unacceptable if we have an available effective treatment we’re not using.
September 8th, 2023
Endless Recertification in Medicine — Some Thoughts About the Tests We Take

William Heath, March of Intellect, 1829.
The tests issued by the American Board of Internal Medicine (ABIM) for credentialing physicians are much in the news again. There’s even a petition circulating to eliminate the Maintenance of Certification (MOC) process entirely, signed by nearly 20,000 physicians.
I have a bunch of memories, thoughts, and feelings about ABIM and the tests they issue. They’re all jumbled around in my head in a formless blob.
So formless, in fact, that I struggled to put them into a well-structured post. So here they are, in 6 chapters, presented in roughly chronological order.
Chapter 1: The Early Days
I remember a meeting we had during my medical residency where someone from ABIM came to our program to announce the end of “lifetime certification,” starting with our year of residency. (I think he was from ABIM … it was a person not on our faculty.) We were the very first year with a new requirement, thank you very much.
No longer would people who passed their Internal Medicine or subspecialty boards be “grandfathered in” for eternity. At 10-year intervals, repeat examinations would be necessary — length, type, cost to be determined.
(Origin of the term “grandfathered” — in case you were wondering.)
The new program was called “Maintenance of Certification,” or MOC, and would provide hospitals and insurance companies a different benchmark for credentialing physicians. You signed up for MOC by sending in a sizable payment, and then the ABIM site would list you as participating in the program. ABIM promised to send you reminders when your 10-year period was about to expire.
As we heard it, it was a decision passed down from above, one with authority that we assumed had strong data behind the policy. We had no way of pushing back or protesting.
No one opposed some sort of process of continual learning for physicians — but wasn’t that the purpose of good Continuing Medical Education (CME)? And it seemed especially ironic that only the older docs got a lifetime pass. We were miffed, but powerless.
The MOC process was widely adopted, and it became a standard part of employer checklists for hiring and retaining medical staff, and insurance companies for keeping or dropping you from their ranks. If Dr. Hugo Z. Hackenbush only passed his original exam, but failed to sign up for the repeat exams, he risked losing his job. Or not getting paid.
Quite the motivation to participate!
Chapter 2: The Middle Years — Criticism, More Requirements, and a Retrenchment
Widespread adoption notwithstanding, the ABIM and its requirements have long had their share of critics. A Newsweek article in 2015 was particularly vicious. More recently, cardiologist Dr. Westby Fisher has tirelessly laid out his arguments for elimination of the MOC requirement (arguments here and here).
Numerous other editorials and position papers have appeared over the years, too numerous to cite, with the message that ABIM has no good data that their process improves physician quality; that the tests are not clinically relevant; that it’s too expensive; that all it does is enrich an already financially thriving not-for-profit organization; and that it fuels clinician burnout.
The apotheosis of these criticisms came when ABIM added layers of other requirements to the MOC process beyond the recertification exams. Remember these “Practice Assessment, Patient Voice and Patient Safety” programs? Yeesh.
We all spent hours completing “quality improvement” projects of dubious (and sometime frankly bogus) worth, just to say we’d done it. Where did all these forms go once we submitted them? Was quality ever improved by these tasks? (Doubtful.) Who reviewed them?
As for “Practice Assessment,” I memorably asked a colleague of mine, a terrific clinical pulmonologist, to complete one of the ABIM mandated surveys on my behalf. He was supposed to call a toll-free number and answer a series of templated questions about the quality of care he’d observed me give to our mutual patients. He kindly agreed to do it.
Or so I thought — he later told me he never got around to it. But ABIM still said I passed his individual “Practice Assessment” standard. Maybe he sent them his response by telepathy?
Under a torrent of criticism, these extra requirements ended in 2015 with a mea culpa from the ABIM leadership. Good move on their part, as it was the very essence of overreaching by a powerful organization. But the recertification process (MOC) remains, as do the criticisms.
In short, complaining about ABIM, and the MOC in particular, offers a rare example of doctors coming together in a remarkable cross-specialty consensus. Accurate quote from Dr. John Mandrolla:
[ABIM] has done what seemed impossible: got docs of all political leanings to agree on one thing —> ending MOC.
Chapter 3: Taking the ABIM Exams
Through volume alone, I’m something of an expert on taking ABIM exams. I have ID specialty training along with Internal Medicine certification, so I’ve now taken the Internal Medicine boards 3 times, and the Infectious Diseases boards 4 times. Lucky 7!
I’ve spent thousands of dollars for the examinations, sitting for a whole day (a work day) in a high-security environment that treats all us test-takers like potential drug smugglers or terrorists. You check in at a test center, surrender all your possessions and place them in high school-style lockers, take the locker key with you, and head to the testing rooms.
These rooms are gloomy, windowless, and airless places, watched over by suspicious test proctors behind one-way mirrors. The rooms are filled with side-by-side desk cubicles. Each desk has a monitor, keyboard, and mouse.
(Computer mouse, not the squeaky rodent with whiskers. All living things would leave this hostile environment as fast as possible, and that includes vermin.)
The testing day has tight restrictions on bathroom breaks and lunch. You can’t ask anyone for help with anything related to test content. (What happened to medicine as a “team sport”?) You have one source of information — UpToDate. You keep feeling as if you’re about to infringe on some unspoken security policy, and end up being reported to the Transportation Security Administration (TSA), putting you immediately on a “no fly” list.
And here’s another thing about those thousands of dollars we pay for the privilege of taking the test — when you finish residency, nobody tells you who’s responsible for the payments. While some have it as a job benefit, most doctors must pay out of pocket, with time away from productive patient care — leading to additional lost revenue for those in private practice or paid by clinical RVUs. If you take a board review course, that’s an additional hefty sum.
In summary, taking the test is no one’s idea of a fun day at work, and the cost of ABIM MOC isn’t trivial — these rankle pretty much everyone I know.
Chapter 4: A View from Inside (Sort Of)
Full disclosure time — for several years, I was on the committee that wrote the questions for the ID boards.
(Ducks.)
I’d meet with ID colleagues twice yearly with diverse areas of expertise, and we’d test our questions against each other with the guidance of an ABIM official. These were incredibly educational meetings, and I learned a ton from my smart colleagues — arguably more than I’d learned preparing for or taking the boards themselves.
We tried to make the test questions as clinically relevant as possible, of course. Part of our job was to weed out those we thought tested pure memorization, or “look ups,” or highlighted outdated diagnostics or therapies. Each question was carefully reviewed so that there was a clear question, and an unambiguous single-best correct answer, at least so we thought.
But — and this is really important — no matter how good the question, we had no idea whether these test questions evaluated the clinical competency of a physician. This was always a big reach.
Two major barriers: First, not everyone in ID has the same spectrum of practice. Even within our little specialty, there are some who focus on transplant ID, and others who rarely if ever see these patients; some do lots of longitudinal HIV care, and others do none; some are deeply involved in infection control, while others barely touch the topic. I could go on and on, but these differences were obvious barriers to writing clinically relevant questions with broad applicability.
And I am 100% sure this issue applies to every medical specialty. Example — Dr. Mikkael Sekeres, who specializes in hematologic malignancies, describes what it’s like taking the MOC exam in Hematology-Oncology:
… over 90% of the questions have nothing to do with the patients I actually see in my specialty practice, and haven’t seen in over 2 decades of practice.
The second problem was the test process itself. To be blunt, nobody practices medicine in the style of the proctored exam — high stress, limited information resources, time-limited, non-collaborative. No testing of the importance of patient communication, eliciting preferences in ambiguous situations, accounting for differences in medical literacy, or the ability to seek out help from other clinicians. In a world of increasingly sophisticated emulations, the ABIM monitored test setting is the polar opposite of practice, and multiple choice question are an overly simplified probe of the complexities of clinical reasoning.
So what is being tested, really, with the ABIM exams? I learned that they’re evaluating the ability of candidates to answer test questions vetted by psychometricians as being “discriminating” — answered correctly by high-performing candidates and incorrectly by low-performing test-takers. These are the best questions, per ABIM standards.
It’s a form of circular reasoning that somehow is supposed to correlate with the ability to care for patients seen in the office or hospital setting. In other words, even our best, most clinically relevant questions sometimes got tossed out as being not discriminating enough — too easy or too hard based on this self-reflecting metric.
If you’re shaking your head right now, I don’t blame you.
Chapter 5: The Latest Innovation — Longitudinal Knowledge Assessment
Astute readers will note that I’ve “only” taken the Internal Medicine boards 3 times. It’s not because I’ve let that certification lapse — it’s because ABIM now offers a new way to maintain board status, and that’s by taking unproctored exams at home, called the “Longitudinal Knowledge Assessment,” or LKA.
Send in your payment, and 30 questions arrive quarterly, giving brief clinical vignettes, a question, and then multiple choice answers. Notably absent from the answer choices is “I would never manage a case like this on my own, I’d ask or refer to a colleague.”
Tough luck. You have 4 minutes to answer each one, and can use any resource.
Whether this LKA process more accurately correlates with clinical competency than the proctored high-security test is anyone’s guess. Four minutes to answer a clinical question? How did they come up with that? Does research show that 4 minutes is some magical threshold that skilled physicians find sufficient for every clinical question? Do the doctors needing 5 minutes need some sort of remedial training in how to look things up more efficiently?
Plus, I have no idea what the criteria are for success in this home testing program. I’m just answering the questions, hoping I’m getting enough questions right to maintain my certification status. La de da.
Certainly this LKA feels less punitive than the proctored exam at the dreaded test center. But there’s a negative trade-off, and it’s not trivial. These questions arrive on a regular basis, an endless 3-month cycle that stretches on indefinitely. It feels like a coach told you to start running laps for fitness but declined to tell you how many.
Big picture — do I really need to do this the rest of my career? I’m only a year or so into it, and confess it feels about as rewarding as renewing your car’s registration or paying your monthly utility bills.
And let the record show that the much-hyped flexibility of the LKA led to one of the most tone-deaf examples of organizational publicity in the history of academic medicine — a doctor posted, on the ABIM site, an account of her doing exam questions while “on the adventure of a lifetime, visiting all the lower 48 States in an RV.”
I’m sure she meant well. But the response from the medical community at the implication that we spend our vacation time doing MOC was predictable outrage.
Never forget!
Your MOC fees paid someone at ABIM to come up with this ad!END MOC sign the petitionhttps://t.co/OYfhsxZkkG pic.twitter.com/MzPI9Ct9Lv
— Aaron Goodman – “Papa Heme” (@AaronGoodman33) August 2, 2023
And while ABIM deleted the original tweet promoting the post (but not the post itself), and apologized for it, some of the responses were very, very funny indeed. Here’s my favorite:
Getting married this weekend and want to get a head start on Q3 LKA questions? Hear from Dr. Gunner, who completed her LKA questions between her wedding ceremony and reception!@ABIMcert pic.twitter.com/VZz18KNQl2
— Ari Elman, MD (@AriObanMD) July 30, 2023
Chapter 6: Looking back, Looking Forward
One of the very best decisions I ever made during college was to spend time doing something else before starting medical school. That one year — spent at a school in England, mostly teaching American literature to kids age 12-18 — was so rich with experience and challenges at my then tender age that it has greatly rewarded me many times over in the coming decades of my life.
Haven’t regretted it for a moment — even though it delayed medical training by 1 year, and hence meant missing out on being grandfathered in on lifetime certification in Internal Medicine. Oh well.
But despite my not regretting this 1-year delay, I can’t help wondering if the ABIM MOC is really the best way to make sure that doctors stay up to date. Couldn’t we do something less onerous? Less expensive? Something tied more closely to the actual clinical practice we do every day, adapted for each person’s particular patient profile? Something linked to existing CME requirements and state licensing? A competing credentialing process, the National Board of Physicians and Surgeons (NBPAS), started in 2015, and has gained some traction. Certainly it has its strong supporters.
In summary, it really does seem like it’s time for a change.
What do you think?
August 18th, 2023
My Vote for the Weirdest Antibiotic on the Planet

Textile Dyeing Machine, 19th Century.
If you’re an ID doctor, there’s an excellent chance you’ve treated patients who have non-tuberculous mycobacteria (NTM) with clofazimine. In fact, based on a poll done with the utmost scientific rigor, it’s well more than half of you.
And if you’re not an ID doctor, there’s a decent chance you’ve never even heard of it — clofazi-what? This is hardly surprising, since it gets my vote as the weirdest antibiotic out there. Of all the drugs we use in ID, clofazimine has the highest ratio of strange qualities to proven usefulness. Plus, it’s fun to say.
Allow me to share why it’s so unusual.
It’s been around a long time.
First synthesized in 1954, it initially held promise as an anti-TB agent, but it didn’t work very well. Later observations found that it was useful for treating leprosy, which is what led to FDA approval in 1969, under the brand name Lamprene. I don’t imagine there was much of a direct-to-consumer marketing effort when this blockbuster was released.
Since in vitro testing also suggested activity against non-tuberculous mycobacteria (NTM) — for which treatment options have always been limited — ID clinicians regularly used clofazimine for years, including both for disseminated M. avium complex (MAC) disease in patients with advanced HIV disease and for pulmonary disease from NTM in immunocompetent hosts.
My first prescriptions for clofazimine were written in the early 1990s for both of these indications. I felt like I’d really arrived in exotic ID Land, writing for this strange drug used for leprosy. Wow.
Despite its approval over a half-century ago, you can’t just prescribe it anymore.
For reasons likely having to do with profit (or the lack thereof), in 2004 the drug disappeared from pharmacies. But it remained an essential drug for the treatment of leprosy in government-funded programs, so the company (Novartis) continued to manufacture it on a limited basis for this indication only.
This put us non-leprosy clofazimine prescribers in a bind. Azithromycin and clarithromycin had come along, greatly improving outcomes for treatment of these difficult bugs. But some MAC isolates and many strains of M. abscessus have macrolide resistance.
Certain enterprising patients heard the drug was still available in Canada (I wonder who told them … hmmm), but that door closed too.
But clofazimine remains widely available in many countries with high TB incidence. One colleague told me it can be purchased in India in most pharmacies for “cents”. It really is striking how often mycobacterial diagnostics and therapies are often more accessible in resource-limited countries than here.
But you can still get it here — warning, the process is not for the paperwork-averse.
Initially, the National Hansen’s Foundation distributed clofazimine on a very limited basis using what’s called a single patient investigational new drug (SPIND) program overseen by the FDA. That changed in 2018, and now Novartis runs this program.
These expanded access programs are in reality clinical research studies — enough so that the clofazimine program appears on clinicaltrials.gov, and approval will be required by a local Institutional Review Board (IRB). That clinicaltrials.gov link contains the critical information about how to get started, which is to email or call the company stating you have a patient who needs clofazimine. They will follow up with a series of essential steps before you can get the drug for your patient — here’s an (old) summary that’s no longer quite accurate but captures the basics.
You certainly won’t find this information on the Novartis site, or at least I couldn’t. Maybe it’s there somewhere.
What’s it like to go through this clofazimine approval process? As the site principal investigator for this program at our hospital, I can attest that Dr. Joseph Marcus provides here a highly illustrative video:
Did a SPIND for one pt at my institution as a first year attending and coordinating everything felt like a final exam/obstacle course of everything I learned in fellowship pic.twitter.com/H1Wc38cr3d
— Joseph Marcus (@JosephMarcusID) July 20, 2023
Hey, there’s a reason why we’re the Paperwork Champs! If it weren’t for the help of our research pharmacy and regulatory coordinator (thank you, Maria and ID pharmacy team!), I never could do this in a million years.
The drug looks active in vitro, but we really don’t know if it works.
Some reference labs will run clofazimine susceptibility testing on NTM isolates and report minimum inhibitory concentrations (MICs), but also add cautionary comments about the lack of standardized MIC breakpoints. In other words, lower MIC numbers seem better — and the results usually are low — but we really don’t know how this correlates with the drug’s clinical activity.
In addition, prospective data on clofazimine treatment in humans are incredibly sparse — and that’s being generous. From an informational slide set provided by Novartis comes this summary table:
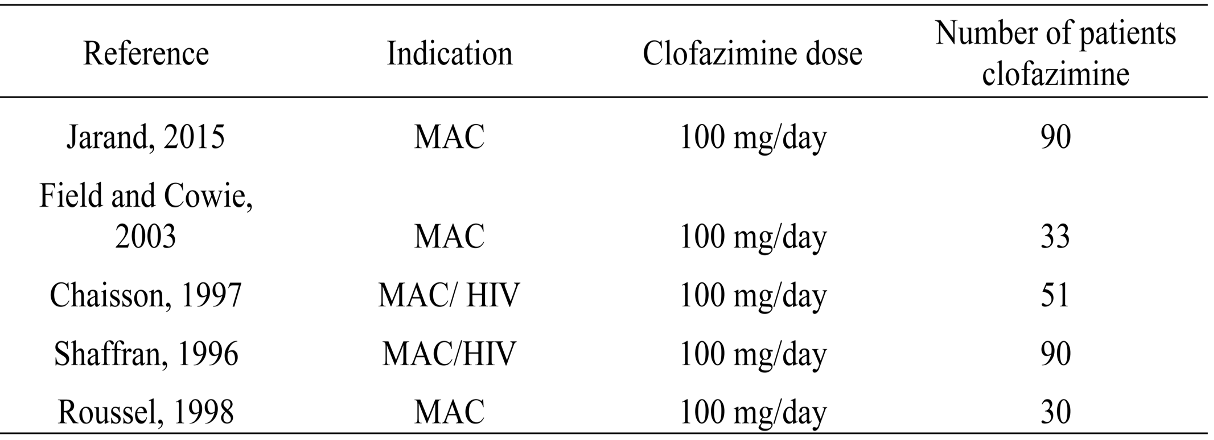
The first study on this slide gives us probably the strongest favorable data — not only does it have only 90 treated patients, but it’s also not randomized and retrospective.
The slide also includes this statement:
No properly designed randomized controlled trials (RCT) have been reported to evaluate the role of clofazimine in the treatment of NTM infections.
Agree 100%. And while there are numerous other published studies besides those on this table, most are case series, not comparative clinical trials.
One possible reason for the lack of good data is the heterogeneity of people who have NTM pulmonary infection, ranging from asymptomatic colonization to progressive, life-threatening disease. Plus there are hundreds of different NTMs, and they can infect other sites, not just the lung.
As a result, this is a very challenging disease to study prospectively. One ongoing clofazimine treatment study opened in 2016; it has a sample size of 102. It still has not been completed.
The end result of this knowledge gap on the efficacy of clofazimine was succinctly put by Dr. Cam Wolf, who wrote:
For these infections, it’s nigh on impossible to tell if clofazimine is the difference maker.
Yep.
But this might change soon. There are other ongoing clinical trials of the drug, with the caveat that most of them involve tuberculosis, not the NTM we more often treat here.
Two randomized clinical trials of clofazimine for MAC in HIV found that clofazimine-treated participants had a higher mortality rate.
In people with advanced HIV disease and disseminated MAC, treatment with clarithromycin, ethambutol, and clofazimine was compared to clarithromycin and ethambutol alone. Mortality was 61% in the clofazimine three-drug group versus 38% in the two-drug group.
(Reminder, the pre-ART era in HIV was grim — and tragic.)
Baseline differences between the two groups, with higher levels of infection in the clofazimine recipients, could explain these unfortunate results — but it also could have been an adverse effect of clofazimine, which has in vitro anti-inflammatory and immunosuppressive effects on both T-cells and monocytes/neutrophils. Could that explain the negative results in this highly immunocompromised group?
A second study in a similar population compared a three-drug macrolide-containing treatment with a four-drug non-macrolide regimen with clofazimine; survival was longer in the former. In this trial, it’s hard to blame the clofazimine, since only the three-drug group received a macrolide, the most active drug for MAC.
Limitations in these studies notwithstanding, we nonetheless generally avoid clofazimine in these very sick people with HIV.
How these discouraging study results inform the proper use of clofazimine in most patients with NTM infection treated today is anyone’s guess. Your typical pulmonary NTM infection case — not immunosuppressed, no dissemination — is worlds away from a person bacteremic with MAC, untreated HIV, and a CD4 cell count <50.
Still, it’s a concern.
We don’t know how it works.
That is, if it works.
But let’s assume it does. The two most commonly cited mechanisms are redox cycling and membrane destabilization and dysfunction. Not particularly strong in either biochemistry or molecular biology, I will share this erudite summary of the latter hypothesis from an excellent review:
This contention is based on the study by Baciu et al., who reported that cationic amphiphiles such as clofazimine partition rapidly to the polar–apolar region of the membrane, where, at physiological pH, the protonated groups on the drug catalyse the acid hydrolysis of the ester linkage of the phospholipid chains. The consequence is the production of a fatty acid and a lysophospholipid, both of which possess antimicrobial activity.
Those are sentences I never could have written. Fortunately, one doesn’t have to understand the mechanism of an antibiotic to use it.
Clofazimine has one common and very strange side effect — and a few others that are less common but very important.
The most famous side effect — or infamous, if you don’t like it — is skin darkening. It occurs in most people, is exposure-related, gradual in onset, and takes on a variety of different hues, from barely noticeable to quite dramatic.
For some, it’s quite flattering, if emulating sun exposure is the desired effect. “My friends all ask me where I’ve been this winter for my suntan,” said one of my patients, as Boston’s winter rays could never produce that warm glow. She likes it.
But for others, it just looks … strange. Watch out if you combine clofazimine with omadacycline (as we increasingly do with treatment of M. abscessus), and add a dose of strong summer sunshine. Covering arms and legs and wearing a broad-brimmed hat are both highly recommended! Fortunately, the skin darkening slowly wears off after the clofazimine is stopped.
Note that the darkening can occur in mucosa, conjunctiva, and body fluids too. Other skin side effects are excessive dryness and generalized pruritus. These dermatologic side effects could be one motivator behind the ongoing study of inhaled clofazimine, which presumably would avoid this toxicity.
Cases of torsades de pointes due to QT prolongation have been reported, as well as a serious syndrome of crystal deposition in intestinal mucosa, the liver, spleen, and lymph nodes. Suspect this crystal deposition problem if a patient on clofazimine develops worsening abdominal pain, and stop the drug immediately. Both the QT prolongation and the crystal deposition are more likely with higher doses.
So there you have it: clofazimine in all it’s extraordinary weirdness, hope I’ve convinced you.
Let’s hope we get some more data on the safety and efficacy of this drug sometime soon — and, if the results are favorable, the hurdles to using it finally come down. Our patients with NTM infection need all the help they can get!
August 9th, 2023
Really Rapid Review — Brisbane IAS 2023

Lamington National Park, Queensland.
You’ll find some conference highlights listed below from the 12th International AIDS Society Conference on HIV Science (or IAS 2023), which took place in lovely Brisbane — where the late July weather was delightful, the ubiquitous ibis was the local nuisance bird, and the riverside parks went on and on and on.
Some might wonder if I can still use the patented, trademarked, and copyrighted Really Rapid Review™ title when I’ve been home from Australia for a whole week. I vote yes — after all, I lost a whole day getting there, and deserve some of that time back. No, my Saturday, July 22, did not exist.
Most links will bring you to the invaluable natap.org site, long may it exist in its delightfully retro late-1990s format.
- Pitavastatin reduced cardiovascular events in people with HIV at low- to moderate CV risk. Yes, I already discussed it in detail, but how could I not at least mention this large, randomized clinical trial? It was the biggest story of the conference, which strangely was put in one of the smaller conference venues. People were literally sitting in the aisles, standing outside, clamoring to get in. Go figure.
- Prolonged ART-free HIV suppression occurred in a person treated with a stem cell transplant from a donor with wild-type CCR5 receptor status. Whether this case will be durable or ultimately have virologic rebound like the Boston patients remains to be seen. “Remission” (no detectable replication competent virus in blood or sampled tissues) is now at 20 months off ART and counting.
- Australia continues to be a model for HIV care and prevention. Their HIV care cascade exceeds 90-90-90% (diagnosed, started on treatment, suppressed), and they also have widely adopted preexposure prophylaxis (PrEP). Anecdotally, some HIV specialists in major urban areas say they rarely see new diagnoses. Imagine!
- 78% of the women in a large study of PrEP chose injectable over oral PrEP after the blinded, comparative phase of the trial ended. HPTN 084 compared TDF/FTC to injectable cabotegravir in women at risk for HIV; cabotegravir was significantly more effective. These results raise the question of whether cabotegravir for PrEP would be more widely used here if logistical and cost barriers were lower — I suspect yes.
- Stay tuned for updated information on prevention of gonorrhea with the meningitis B vaccine. In the ANRS DOXYVAC study, the factorial design randomized participants to doxycycline PEP and/or the meningitis B vaccine or neither. Though both interventions were reported at CROI 2023 to be significantly protective against gonorrhea in the interim analysis, investigators observed a discrepancy between the interim meningitis B vaccine results and the complete analysis — hence we await review and reporting of the final results.
- At Fenway Health (an LGBTQ Care Center in Boston), they have started using doxycycline post-exposure prophylaxis (PEP) to prevent sexually transmitted infections (STIs). I suspect most of us have also begun offering this (I have), in particular to those with recent bacterial STIs. Guidelines endorsement coming soon, I hear.
- At 48 weeks, doravirine/islatravir was non-inferior to bictegravir/FTC/TAF as initial therapy. One versus zero participants developed treatment failure and resistance. No differences in weight gain (roughly 3 kilograms). Because the 0.75-mg daily dose of islatravir was used, CD4 response was blunted in that treatment arm. This comparison is now being repeated with 0.25 mg/daily islatravir dose; it will be interesting to see if virologic responses are as good.
- The key doravirine resistance mutations occurring in prospective clinical trials were V106A/M and F227C/L/R. These aren’t the only mutations that lead to doravirine resistance (notably Y188L and M230L do as well), but in the rare patient with virologic failure on doravirine-containing regimens, these two are the most likely to occur — and they don’t lead to cross-resistance with efavirenz or rilpivirine. Memorize these mutations, impress your friends!
- DTG/3TC was non-inferior to DTG + TDF/3TC in treatment-naive PWH, even without baseline resistance testing. 30% had HIV RNA >100,00, 21% CD4 <200. No treatment-emergent resistance. The study was slightly underpowered (n=214), but it highlights how rare transmitted 3TC or DTG resistance is, fortunately enough.
- People with virologic failure on DTG-based three-drug regimens had a high level of resuppression after adherence counseling. The exceedingly low rate of resistance selection with these DTG-based regimens likely explains the favorable results, which obviate the need to switch treatment. Suspect strongly these results apply similarly to BIC/FTC/TAF treatment, at least based on similarly low incident resistance and anecdotal experience.
- In treatment-experienced persons with HIV (PWH) with a history of treatment failure but current virologic suppression, switching to DTG/3TC was successful even with known 3TC resistance. 50/100 participants had documented M184V mutations. These results notwithstanding, until there’s a fully powered study evaluating this question — and why would we do that? — my view is that DTG/3TC has been extensively tested in people without M184V, and this is the population best suited to this switch strategy. By the way, this study is called SOLAR-3D, not to be confused with the SOLAR study of CAB-RPV.
- Switching stable PWH with integrase-inhibitor weight gain to darunavir/cobicistat/FTC/TAF did not reduce weight. Right now, it’s looking like the only regimen switches that may induce weight loss must involve a TDF-based treatment — which has its own toxicity issues (renal, bone), especially in older patients.
- For treatment of hepatitis B and HIV co-infection, BIC/FTC/TAF and DTG + TDF/FTC were both effective. These 96-week results continue to show certain endpoints favoring BIC/FTC/TAF, most notably HBEAg loss and seroconversion. But the DTG + TDF/FTC arm has “caught up” to the BIC/FTC/TAF arm in hepatitis B viral suppression, which happened faster in the TAF treatment arm.
- In a randomized trial comparing BIC/FTC/TAF to CAB-RPV, patient-reported outcomes favored long-acting injectable treatment. These results recapitulate several prior analyses with similar findings, which show that if people want to take injectable therapy, they’re happier once they do so. We could interpret these findings as either reassuring or a “self-fulfilling prophecy” — but how about both? And this, by the way, is the other SOLAR study.
- In a heterogeneous population (735) of PWH with viral suppression on three-drug therapy, switching to DTG plus either 3TC or RPV was safe and effective. Unlike in the phase 3 studies of these treatments, 50% of the population had a history of drug resistance mutations, so this expands the population potentially eligible for using two-drug therapy. Assume (hope) that they did not have primary mutations to either of the two drugs in the given strategy!
- In a small, comparative, matched study, PWH and diabetes mellitus (DM) lost significantly more weight on GLP-1 receptor agonists than people with DM alone. Our limited experience in clinical practice certainly shows this class of drugs leads to weight-loss in PWH. Could GLP-1 agonists be particularly effective in our patient population?
- In treatment-naive people starting antiretroviral therapy (ART), DTG-based regimens were associated with incident hypertension, especially when combined with TAF. This analysis from the NAMSAL and ADVANCE studies also showed that hypertension treatment (available to the ADVANCE study participants) was quite effective. As noted here and in a bunch of other studies looking at hypertension and association with specific ART, it’s tricky to disentangle this effect from differences in weight gain — a well-known trigger of high blood pressure.
- New onset diabetes was associated with current integrase strand inhibitor (INSTI) use, even when controlling for BMI. As noted by some HIV specialists, this same cohort found an association between INSTI use and cardiovascular disease — an association later refuted by an analysis controlling for baseline CV risk factors.
- Bictegravir exposure was reduced in pregnant women taking BIC/FTC/TAF during the second and third trimesters. The median trough was still 6-7 times higher than the inhibitory quotient, and none of the women experienced virologic rebound or failure; the only participant with notably low levels was taking concomitant calcium and iron supplements. These results support the decision to continue BIC/FTC/TAF in women who are found to be pregnant while receiving this regimen.
That’s a wrap! What did I miss?
After the conference, we went hiking in Lamington (see picture above) and Springbrook National Parks. So beautiful! Saw wallabies and cockatoos and kookaburras!
Now, if only someone could come up with a cure for jet lag …

