An ongoing dialogue on HIV/AIDS, infectious diseases,
April 25th, 2022
Yes, Relapses After Paxlovid Happen — Now What?

Young Monk in the Greek Style, from Mascarade à la Greque, Alexandre Petitot (1727-1801).
Around two weeks ago, one of my long-term, very stable patients with HIV called me saying she’d just been diagnosed with COVID-19. Over 60 with hypertension, and overweight, she qualified for nirmatrelvir/r (Paxlovid) under the Emergency Use Authorization (EUA), and took it without problem.
(Certain details changed for confidentiality.)
In fact, she started to improve within 24 hours of the first dose. Complained a bit about the metallic taste, but was thrilled at this rapid recovery.
She then contacted me again a week later, saying she’d relapsed. More nasal congestion, cough, and fatigue — not as bad as when the illness started, but unmistakably a relapse. Not only that, the home antigen test, which started out as a dark line at the outset, became a barely discernible line after the treatment, and now was clearly positive again.
Her biggest concern was getting back out in the world without infecting someone. She really wasn’t that sick; she just wanted advice about when she could return to work and start socializing again.
“Avoid close contact with others until that test clears,” I said. (Which it did a few days later, and she completely recovered.) But certainly the whole thing was a head-scratcher for both of us.
Turns out these relapses do occur; this was just the first time I saw it. The investigators from Pfizer observed them in their clinical trial EPIC-HR, and reported it to the FDA. Take a look at this figure from their study, which was not included in the NEJM paper, but did appear in an FDA report:
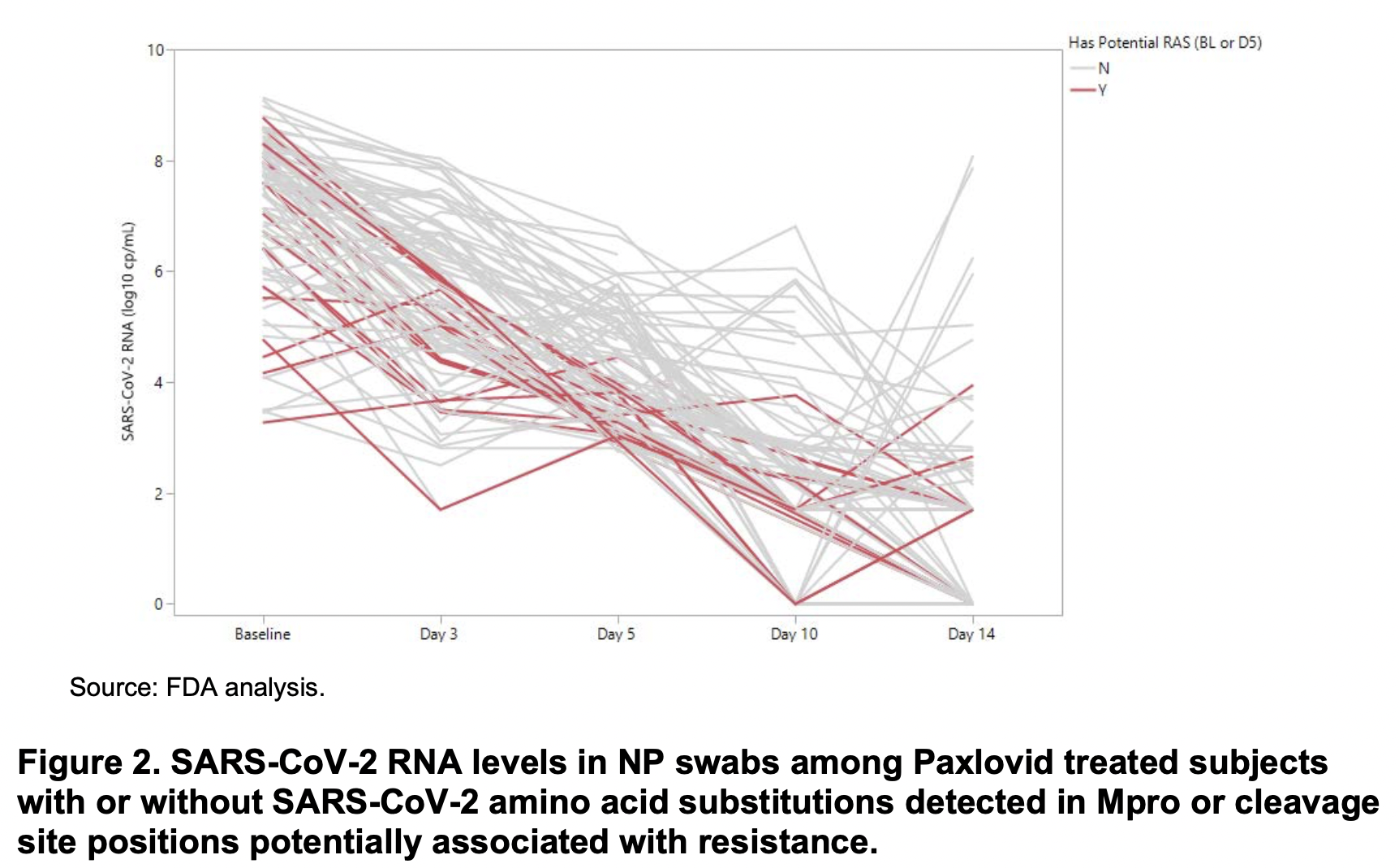
See those lines heading upwards from day 10-14? Those are relapses. Sequencing of SARS-CoV-2 from these cases did not demonstrate resistance mutations either at baseline or at relapse that correlated with resistance. Specifically:
In summary, currently there are no clear signals of baseline or treatment-emergent NIR resistance from the preliminary analyses of clinical trial EPIC-HR. These analyses will continue to be conducted as more complete data from EPIC-HR are obtained and reported.
A detailed analysis of a similar relapsing case is available as a pre-print — viral sequence the same at 3 time points (no resistance or reinfection), respiratory multiplex PCR negative (no new pathogen), good antibody response.
Not surprisingly, as use of Paxlovid increases along with the supply, the anecdotal reports of these relapses increase in parallel. Not just in clinical practice — they’ve popped up on social media and in the press. It’s now become a standard part of my counseling to people, that this return of symptoms and test positivity might happen.
So we know that relapses happen. What don’t we know about these cases? Quite a bit, actually.
- How often does it occur? It’s tricky from the figure to get a clear picture of the numerator and denominator. And of course, that was in an unvaccinated, high-risk population. What about those who are vaccinated?
- What are the risk factors? Could it be that those with baseline high viral loads/low cycle thresholds are at greater risk of relapse? People who are severely immunocompromised? Older? With some variants more than others?
- Does antiviral treatment blunt a helpful immune response? Does the immune system need to see a certain concentration of viral antigens to provide adequate clearance? Or are those who are relapsing just the subset of people who would have had prolonged viral shedding to begin with?
- Should we assume that people who relapse become contagious again? That’s my assumption — and it seems highly likely — but it’s worth proving in a research lab that these viruses are just as replication-competent as the pre-treatment viruses.
- Does the virus develop resistance during this 5-day course? So far this hasn’t been reported in a relapsed case, at least a far as I know. Seems inevitable at some point, however, so worth looking for, again in the context of a research study.
- Should treatment courses be longer? Maybe a longer course is better, but maybe not. How about a 5- vs. 10-day blinded clinical trial, with clinical, virologic, and immunologic endpoints? (Importantly, both 5- and 10-day courses follow the rules.)
- Does this happen with other antiviral strategies? I don’t recall hearing similar patterns with remdesivir or molnupiravir, but then again viral load reductions were less robust. This could be related to the mechanism of action of viral protease inhibitors.
- Should highly vulnerable people with relapses be treated again? Somehow “do nothing” for the most at-risk people with COVID-19 (for example, those on rituximab) doesn’t seem right when we have treatment tools at our disposal. One of my colleagues pointed out to me that neither repeated courses of nirmatrelvir (something I suggested in the above-linked Boston Globe piece) or treatment with molnupiravir would be allowed under the EUAs, as they must be within 5 days of symptom onset. It’s likely also that the bebtelovimab window of treatment (7 days) would be exceeded. That leaves 3 days of IV remdesivir as the only outpatient treatment option, which is very hard to access.
Given all these unknowns, it would be enormously helpful for Pfizer to release further data on their relapsing cases. Not just how often they happened, but also how they did clinically — presumably they did well, given the overall favorable results from the study. Any further information on immune responses? And did they paradoxically have a longer time to viral clearance than the placebo arm?
Fortunately, I’m aware of several research groups who are studying these cases right now here in Boston, and undoubtedly others are doing so elsewhere. We should know more soon.
But what we do know now — and I’ll keep saying this again and again — is that this is one tricky virus, full of surprises.


Do we know anything about the relapse rate without paxlovid? Wondering if there is some self-selection bias at play with the relapse reports. Only a person who gets tested can take paxlovid, and only a person who tests could confirm a relapse. Whereas, someone who doesn’t test could have relapsed and just assume it is a separate, unrelated infection.
I just got a case of rebound COVID after taking Paxlovid. I got better quickly, was fine for several days, tested with a faint pink line, then got really really really sick! I tested again, and the line was hot pink again. I’ve been so for three days that I was thinking it was the worst cold in history. Now I learn it’s not a cold at all.
And it’s not so rare. My sister-in-law is going through the EXACT thing this moment, and we caught COVID from our elderly mother-in-law, who gave it to our husbands and us.
I am in the exact same boat here- and it’s been 5 days since I rebounded and started testing positive again. When does this end? How long did it last for you?
FWIW, early 60s, healthy, vaccinated and boosted. Took Paxlovid starting day 0. Symptoms mostly clear by day 2, felt fine by day 5 or 6. Mild symptoms back on day 11 with a strong + on a RAT.
Any information from Pfizer about what happened with those participants after day 14?
I took AV after testing positive on 19th April. Was negative by 25th and felt fine! Woke up this morning 29th – feeling dreadful (very bad cold symptoms) and I’m very firmly positive again. Rang Pzfizer directly who had no answers! (I’m in the UK)
I took the course of Paxlovid and had a relapse myself. Initial symptoms were dry cough and congestion. Not too bad at all. Was relieved for sure. During the first break, before relapse, tested antigen negative twice, I felt no lingering symptoms and had returned to very normal active life including exercise etc. Then relapse after 9 days symptom free, and 2nd round was at least as bad as the first time, and now I have a lingering dry cough and tightness in the chest. It’s been five days since I tested negative the second time. Would love to know more or participate in any studies if need be.
I didn’t realize this was a phenomenon when I was prescribed Paxlovid. My initial symptoms included a high resting heart rate when I tested positive of 4/13/22. Began the course of Paxlovid the following day and began feeling better on the 4th day of treatment. Everything improved and my tests were eventually negative. On 4/26, just over a week after stopping the Paxlovid, my resting heart rate again spiked over 100bpm. Then, my cough returned and I confirmed a new positive antigen test today.
Tested positive, and got the antiviral treatment. Paxlovid. No risk factors except my age which is 72. Felt better very quickly, within 2 days. Finished the 5-day treatment course, and about 5 days after finishing, symptoms came back. Tested strongly positive again. It’s been about 5 days since the onset of symptoms the second time, and I’m still feeling pretty bad, actually worse than the first go around. Starting to think that the paxlovid was a bad idea, at least for me.
I am one of these cases as well- all the comments above match my experience. Is there anyone we can report this to at Pfizer or the FDA? And when did people start to feel better? I tested positive again (after 3 days of being negative) five days ago. I feel horrible today and would love some relief. I would also love some guidance on whether or not I am still infectious at day 17 of this crud. No one seems to have any answers.
I’m glad for these posts to realize that my experience is not as mysterious as I initially thought: I got moderately miserable constitutional symptoms and a harsh cough on Day 0, rapid COVID-19 Ag test was negative that day, but positive the next. I started Paxlovid on Day 1 and was afebrile by day 2. Dramatic improvement. I completed a 5 day course and felt truly normal by day 7-10, at which point mild cough and fatigue recurred and my rapid test was strongly positive again (?still).
Yesterday, day 12, a very runny nose began, continues today, and I just feel like I have a bad cold, responding well to symptomatic treatment. My rapid test is still strongly positive, so I’ll continue to isolate and test, rest and treat symptoms. Thanks for this reporting!
Forgot to mention, I am vaccinated and boosted x1, 5 months ago – was just planning to get booster #2, which I will now put off for a while I think.
Thank you for bringing this issue into the light; I’m experiencing the same thing. I am a fairly healthy 51 year old male with HTN and fully vaccinated with one booster at the end of October. I tested positive initially with a RAT at home on 4/21 after feeling tired and having a mild sore throat the evening of 4/20 which by the next day included cough and congestion. I started Paxlovid on 4/22 or Day 2 and continued to test positive until Friday 4/29 Day 9, when I ended isolation and began interacting with my household again/began leaving the house with a mask. I felt much better until the evening of 5/2 Day 12 when I got a fever and cough came back, and this morning Day 13 I tested positive again on RAT. This must be studied–I am concerned my family has now been exposed since per CDC, I was past 10 days, tested negative, and stopped wearing a mask in the house.
My experience exactly. Day 2 I tested positive via home test. Day 3-8 paxlovid. Did great on it, and afterwards, though a bit winded. Through day 12 fine. I was celebrating the beauty of paxlovid. Then day 14 I had terrible head congestion, dry cough, exhaustion… worse than onset. Still raging day 17. After reading this I decided to test again: Positive. Now I’m so wishing I tested when I felt good. But we will never know. I was around a ton of people days 10-17, unmasked. Was I contagious? Grandkids are coming tomorrow. Should they? Thinking “no.” Not sure when I am safe to be around others, or when I might feel better.
I sent this email to the FDA drug evaluation group yesterday. Later yesterday, I tested negative again on a RAT, and I feel better but still some congestion this morning. This blog is the first good summation of the issues at hand I have seen. Thank you Dr. Sax.
To Whom It May Concern:
I am a surgeon. I began mild upper respiratory symptoms early morning 4/21. While I had no known exposure and am exceedingly careful, because I needed to attend my stepson’s funeral the following week, I used a home test. I was surprised to find it positive for Covid. I am 65 with some risk factors for more severe disease so I took a 5 day course of Paxlovid beginning at 2pm on 4/21, completed around 11pm 4/25. During the 5 day course, I felt essentially well with resolution of rhinorhea and sinus fullness but developed a moderate pharyngeal pain with swallowing, left greater than right. Curiously, this was odynophagia, not a sore throat from mucosal inflammation. This waned and was resolved completely by 4/26. My husband remained test negative & asymptomatic throughout. I retested 4/28 morning before leaving for the funeral and was negative. The burial 4/29 was 2 hours outside in 40 degree temperature with 30+mph winds. About 8 hours after that, I began to have rhinorhea, sinus fullness, sneezing fits, cough and malaise. We were driving 7 hours home, I tested at midnight on 4/29, around 5 hours after symptoms began again, and I was once again positive for covid. I have been searching for information about this rebound effect but find only media reports echoing one another. None of my colleagues knew what to recommend (retreating? resume isolation? When to count the 10 days?). There is absolutely no guidance here. I chose not to retreat and am toughing it out, reasoning that 1) it just seemed to kick the can down the road and 2) perhaps treating would decrease any immunity benefit from having a mild to moderate case of covid. I will say, the rebound symptoms were far worse than the initial case. Perhaps this is because I treated so promptly? But on 4/29 & 4/30 I felt flushed, tachycardia to 98 (usual heart rate 60-70s), significantly worse upper respiratory symptoms but no return of the odynophagia. Today, 5/3, I am feeling better but still have excessive mucus production and a hoarse voice. My husband remains negative by symptoms and by antigen tests.
I could find no official avenue to report this case so I am sending this email to you in hopes that it will help inform the evaluation of Paxlovid treatment. I chose to continue to quarantine at home and will retest when symptoms are further resolved. If you do have any data about this, I would appreciate any information you can provide.
Sincerely,
Dr. Lynn M. Smolik
Dr. Smolik,
I have updated some of the information we’ve received from Pfizer and the FDA in my latest post.
Thanks for commenting.
– Paul
https://blogs.jwatch.org/hiv-id-observations/index.php/more-on-relapses-after-paxlovid-treatment-for-covid-19/2022/05/04/
Should add, both my husband & myself were fully vaccinated with Moderna, initial doses Jan-Feb 2021, 3rd dose (100 micrograms) Sept 2021.
I’m on day 2 of Paxlovid and after reading this article and comments, I’m thinking maybe I should discontinue it and let my own immune system fight the virus. It sounds like the Paxlovid is “burying” it to the point where my immune system isn’t engaged. Wouldn’t it be better to let my own immune system do it’s job? Thoughts?
If the variant is more infective, then it may still be infective at a higher Cycle Threshold.
For example, it may still be infectious above at a CT of 4o or 50.
Maybe the CT cutoff for a negative should be at 40, or even 50
Isn’t anybody concerned that these folks who are unable to clear the infection despite the Paxlovid will be seeding the next variants? Not only that, but that this will create a selective pressure for treatment- and vaccine-resistant variants?
Karen, maybe look at optimizing your vitamin D levels and taking some zinc to get over the COVID infection (either along with the Paxlovid or instead of it). Lots of studies confirm the essential role played by vitamin D for the immune system, and zinc clearly combats respiratory viruses (add vitamin C and quercetin (which drives the zinc into the cells without the need for heavy doses) for an added punch)). Wishing you good luck and a speedy recovery!
Another rebound case here. 41 year old moderetly healthy male Vax and boosted with Moderna. First symptoms on 4/25. Tested positive 4/26. High fever, cough, and congestion. Took paxlovid starting the night of 4/27. Felt better after a couple days with the exception of mucus and began testing negative on RAT 5/3 and PCR on 5/4. Symptoms re-emerged the evening of 5/7 with a full on cold consisting of mostly of a runny nose, fatigue, and sneezing in 5/8. Tested negative in RAT 5/8 but positive on PCR 5/8. Tested strong positive on RAT the morning of 5/9. So far symptoms feel a lot milder and no fever (knock on wood).
I am having the exact same experience as many describe on this site. I treated with Paxlovid and recovered very quickly from the initial infection, and now I have rebound symptoms (12 days after initial infection and similar to the common cold.) It is now day 3 of the rebound, and I am wondering how long it is going to take to fully resolve. Ugh!
I’m on day 4 of Paxlovid. Can I stop taking it? Or do I have to finish the 5-day course of treatment?
I tested positive on April 27. A pink line showed up a few minutes after I started the test. I started Paxlovid that evening. My symptoms (which were mild to moderate) were resolving but not gone when I tested negative on day 6. I masked and did errands. My residual symptoms, mostly a sore throat and hoarseness, continued to improve. On May 8 my glands felt slightly swollen and my nose was very runny. I tested very strongly positive, much more so than initially. My symptoms were extremely mild. I reported this information to my PCP, figuring she would want to know since this is a new drug. Her discouraging response was that she doubted the accuracy of the test! I quarantined, tested ever so slightly positive on day six and negative two days later. I’m continuing to mask per guidelines and feel fine. I would be happy to participate in any study.
Joining the crowd. 45 yo female, fairly healthy but with asthma and IBS, and often viruses lead to bacterial infections. Symptoms 5/1, RAT+ 5/2 with cold symptoms. led to shortness of breath, cough, congestion. Paxlovid 5/4-5/8, symptom resolution other than fatigue and brain fog, RAT- 5/9 and 5/10. On 5/13, day 5 after finishing Paxlovid, began to feel sick, tested, faint positive. Minor cold symptoms but dark positive test 5/14-5/16 and today return of sore throat, chest tightness, and fever! So this is day 16 since first positive, and day 5 since relapse, and been getting worse not better. How long did it take people to recover from a symptomatic resurgence after Paxlovid??
My husband, a 63 yr old with type 1 DM (diagnosed 6 mo. ago- well controlled and he is otherwise active and healthy). Started Paxlovid on day 1 after symptom onset. Symptoms resolving on day 3, negative antigen test on day 8, Symptoms returned day 15. Antigen test very positive. He only had symptoms for a day- but stayed positive for a week ☹️. I’m glad he had such a mild course, so I think it is definitely worth it- but those two isolation periods were a pain. I’ve heard people are possibly more likely to relapse if it is started very early in the infection. It will be nice when they can get the timing/length of treatment figured out….
I am a 67 y/o retired internist who is on prednisone and methotrexate for sarcoidosis. I am fully vaccinated with 4 doses of Moderna vaccine, and have also received a dose of Evusheld in March. I started getting sick with low grade fever, sore throat and cough on April 15, had a positive rapid antigen test on April 16, and started Paxlovid that day. My symptoms improved, but never resolved, and antigen tests done every several days remained positive. 2 weeks later, I had a clear rebound in my symptoms. I am now 33 days out from the onset, still have mild cough, congestion, and fatigue, and my rapid antigen tests (done every 2 – 3 days) remain positive. The lack of guidance and study on immunocompromised patients is disheartening!
I agree. My son has sarcoid. More studies are needed. Paxlovid doesn’t seem to be a cure at all. I would not take it.
My GP recommends ivermectin 5 days along with Azithromycin, & Hydroxy. I am just getting over Covid, these drugs helped me.
My husband and I both tested positive shortly after Mother’s Day, we took Oaxlovid as prescribed and felt better. On May 20th, we both started with cold symptoms and tested positive again. (retired MD and RN) This is not 1-2% relapse?