An ongoing dialogue on HIV/AIDS, infectious diseases,
January 4th, 2021
Ivermectin for COVID-19 — Breakthrough Treatment or Hydroxychloroquine Redux?
It’s an indisputable fact that we need better treatments for COVID-19.
This is particularly true in the outpatient setting. Let’s count how many we have today, hmm, this shouldn’t take long. That would be zero — the same number we had over a year ago, when the disease first emerged in China.
Something safe, easy to take as a pill, and inexpensive. Something that isn’t a costly bioengineered molecule that requires lengthy infusions, given within a short time after diagnosis, to a highly contagious person.
Enter ivermectin — and let the controversy begin.
Yes, ivermectin — the drug licensed for use against strongyloides and other parasites, and probably best known to primary providers for its off-label use for scabies and head lice, and for pet owners as a common de-worming agent.
Of course upon hearing about this “repurposed” antiparasitic drug, many will develop a weary feeling of deja vu.
Didn’t we make this mistake with hydroxychloroquine? Wasn’t excess enthusiasm for this treatment a dismal chapter in our clinical and research approach to this new disease? Enthusiasm that led to wasteful, duplicative clinical trials, flawed observational studies, and irrational prescribing with arguably more harm than benefit? Harms that included not just side effects, but also drug shortages and pointless stockpiling?
Yes to all of the above.
Note that the harm done by the hydroxychloroquine controversy continues to this day. I know investigators who led well-done, fully powered clinical trials with negative results who were, and continue to be, viciously attacked in emails and on social media. Accusations typically charge that they succumbed to pressure from big pharma, or obscured favorable study results based on political agendas, or both.
Believe me, they wanted hydroxychloroquine to work. We all did! It’s relatively safe, inexpensive, widely available — but oh well, the clinical trials showed us it didn’t do much of anything.
Now — back to ivermectin. Where do we stand today? Unlike in the spring, when hydroxychloroquine use was rampant pretty much everywhere (and even appeared in some institutions’ treatment guidelines), this poll suggests that ivermectin use now is much more restrained:
Hey #IDTwitter and other clinicians who care for people with #COVID19. Have you prescribed (or recommended) ivermectin for this disease? Please vote and comment. Thank you!
— Paul Sax (@PaulSaxMD) January 3, 2021
However, there is a strong likelihood that the group voting on this little poll does not represent clinical practice globally. An article in October cited widespread use throughout Latin America, and many commenters to the above poll mentioned similar practices in their countries. Here’s a group of mostly South and Central American clinicians strongly advocating for ivermectin therapy for COVID-19.
Furthermore, many push for broader use of ivermectin in the U.S. as well. A group called the Front Line COVID-19 Critical Care Alliance — made up of predominantly critical care clinicians — devotes much of its organization’s homepage to ivermectin’s promise for COVID-19 treatment, which they summarize in enthusiastic detail here.
Beyond these accounts, what else is out there?
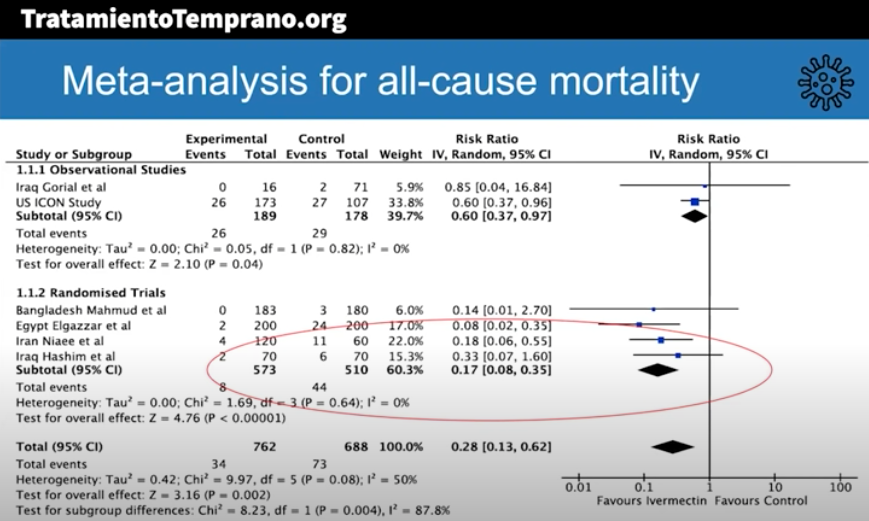
The best summary of the research evidence to date appeared recently in a presentation by Dr. Andrew Hill from the University of Liverpool. In a conference hosted by MedinCell, a company developing a long-acting ivermectin preparation, he presented the interim results of a meta-analysis funded by Unitaid.
Here are some key results (posted with permission):
The risk-ratio for mortality with ivermectin was 0.17 (95% confidence interval 0.08, 0.35), an 83% reduction in risk of dying. Outcomes for other endpoints (time to viral clearance, time to clinical recovery, duration of hospitalization) also favored treatment over controls.
Andrew was kind enough to speak with me today, mentioning that additional clinical trials will be included in the final meta-analysis, and that results are confirmatory. Some studies also included inflammatory markers such as D-dimer and IL-6, with favorable outcomes seen in these endpoints as well.
The presentation appropriately cites the limitations of the meta-analysis, which include the incompleteness of the data, that some of the studies were open label, and the difference in dosing regimens and endpoints. Also — critically important — publication bias may play a role, where we’re only privy to the studies that have what he calls the “good news.”
To further muddy the waters, “publication bias” is a generous term — none of these trials have yet been published in peer-reviewed journals.
Doubters will also understandably cite the very unimpressive pharmacokinetic (PK) data on ivermectin’s antiviral activity. Here’s our ID PharmD Jeff Pearson’s take:
I hope I’m wrong, but I don’t believe ivermectin will work for the treatment of COVID-19 for a variety of reasons (mostly being its PK, see here, here, and here).
But of course PK studies don’t always correlate with clinical activity, and ivermectin may have anti-inflammatory activity, as shown in this animal model.
My take-home view? The clinical trials data for ivermectin look stronger than they ever did for hydroxychloroquine, but we’re not quite yet at the “practice changing” level. Results from at least 5 randomized clinical trials are expected soon that might further inform the decision. NIH treatment guidelines still recommend against use of ivermectin for treatment of COVID-19, a recommendation I support pending further data — we shouldn’t have to wait long.
But we have to guard against two important biases here. First, that because we were burned by hydroxychloroquine means that all other repurposed antiparasitic drugs will fail too.
Second, that studies done in low- and middle-income countries must be discounted because, well, they weren’t done in the right places.
That’s not just bias, it’s also snobbery.



Dear Dr. Sax,
I thank you for your fairminded analysis. Question for you: the level of evidence you are calling for in order for the NIH to stop “recommending against” ivermectin – from what precedent do you draw this? The majority of routine practice as well as professional society recommendations come from lower levels, and that’s during nonpandemic times. A 2010 analysis published in Clin J Inf Dis revealed, for example, that close to 85% of IDSA recommendations are based on observational studies or expert opinion only. Only about 15% drew on even a single RCT. Also, two landmark 2000s NEJM studies demonstrated high concordance of reproducible research loweer on study hierarchy, and larger double blind placebo controlled trials. Is a pandemic the time to INCREASE the evidence threshold to cautiously adopt a practice? If you are against liberalizing it I understand, but why “conservatize it” especially with an inexpensive, safe, unburdensome option, and our health systems stressed to a near breaking point?
My concern, that I think will require a regulatory do’s is thus:
The US Vaccines are authorized for use under an Emergency Use Authorization. such an authorization is contingent upon there being no safe, effective treatment available. Acknowledgement of Ivermectin may put the EUA governing the vaccine distribution in legal jeopardy. I welcome other thoughts on the matter.
HCQ has of course been shown to be effective in early treatment of covid in dozens of studies: https://c19study.com/ HCQ has well known anti-thrombotic and immunomodulatory properties, both of which are highly relevant to prevent severe covid if applied early (outpatient) in high-risk groups. The Recovery and Solidarity trials administered toxic doses in ICU patients, which is irrelevant. Other trials looked into infection prevention, also irrelevant. Yet other trials studied low-risk populations, also irrelevant. But Ivermectin does indeed look even more promising, as it appears to work even in prevention and in late-stage disease.
Glad to see this. Agree we should be scrutinizing available quality data, since early and outpt therapies are needed. Thanks!
https://ivmmeta.com/
The Society of Infectious Diseases Pharmacists has an excellent video on the topic: https://youtu.be/SokdEQXq2AQ by Kati Shihadeh, PharmD, BCIDP worth checking out
The video is gone. Another Ivermectin video was taken by Youtube. Why?
Paul,
Nice review and I agree with many of your points. In particular, we should treat RCTs in developing countries with the same attention to detail we would use if they were done in Boston. That said, much of the data in the meta analysis suffers from lack of blinding, lack of detail about randomizaiton, small size, lack of clearly defined endpoints. A meta-analysis of badly flawed data does not strengthen the data….
That said, I also agree there is a lot more possible signal than there ever was for hydroxychloroquine, zinc, vitamin D, etc. I am a fan of high quality observational studies, but in COVID they have really underperformed, perhaps because the unmeasured confounders are really important.
At least one adequately sized, well designed, double blind trial of ivermectin is urgently needed.
I am absolutely perplexed by the level of hate for repurposing the old, safe and cheap medications to treat Covid . Studies using Ivermectin are pretty clear that is reduces hospitalization ,when used early , and reduces mortality in hospitalized patients. When we currently have nothing to offer the non-hypoxic patient . Why not try it . I ask what do you have to loose
I Read the flccca group compilation of studies (about 100 references) and lisented to Dr Kory’s testimony. I think he’s a straight shooter and exhausted ICU doc. As a 70+ doc on 3 bp meds, I will certainly take it if my family gets covid. The risk and cost is so low and the odds of benefit reasonable though not yet 100% proven. Shouldn’t the NIH be issuing no recommendation rather than recommending against it in this bad winter before Vaccination starts winning. If there is benefit shown by spring in perfect studies, how many folks will die because of their suppression of use. The Argentina study for prevention with 800 health workers on drug with 0 covid vs 400 on placebo with 58% covid seems beyond selection bias so I vote for more tolerance for use this winter while waiting for the perfect studies to be peer reviewed in the west. Thank you
SAFETY first, then EFFICACY ! Just like the vaccine.
I am also a 70 plus MD still seeing patients. I took Ivermectin and I use lots of colloidal silver (don’t drink it). I’ve been exposed several times. Still negative weekly (blood test).
I’m an “n of 1” but so is each relative and friend I’ve recommended colloidal silver to. None who use it has gotten COVID !
Our oath is “do no harm. “ FDA and NIH have no data that prove Ivermectin or silver have anything but a good risk-benefit ratio. “Papa Smurf” (the blue man FDA and big Pharma keep referring to) died of heart disease and not from silver toxicity. Furthermore, both Ivermectin and silver are CHEAP !
Dr. Hartz.
How do yo use colloidal silver?
Thank you.
There are several signals supporting well-controlled, randomized, clinical trials with hard endpoints for ivermectin. But the efficacy of an intervention can not be proven with signals or meta-analysis, particularly when the latter omits quality grading of the studies included in the analysis, Until we have results from the ongoing controlled studies, I see no reason to reconsider the recommendation against the use of ivermectin for treatment of COVID-19.
The largest RCT compared it to HCQ- which has been shown to increase mortality – so what does that mean? Full dose A/c in ICU trial paused for harm potential https://www.nih.gov/news-events/news-releases/nih-activ-trial-blood-thinners-pauses-enrollment-critically-ill-covid-19-patients so MATH may have been right on steroids but not A/c.. Lets all push for large RCT and respect the rule of first do no harm. To quote Paul Marik from his incredible single author text book on critical care”.. If a study seem too good to be true, it is likely too good to be
true” Interesting that ivermectin is so cheap yet company developing a proprietary formulation. Perhaps we should try to get philanthropy money to do Recovery or Solidarity type and get a real answer in large RCT
How about the strongyloides hypothesis?
All commenters and Dr Sax make sense using cogent epidemiologic principles and clinical trial best practices. Dr.Osgood points out that many, if not most, professional orgs use levels of evidence based on observational studies and expert opinion. Dr. Swanson makes a very powerful argument as well regarding the total lack of any out-patient therapeutics (as did Dr. Sax) for early disease. We have nothing! Given that, why haven’t specialists in the West, if they consider other countries research re Ivermectin suspect, rallied to use Ivermectin in smaller, more easily designed proof-of-concept studies? These are studies that are cheaper and more easily crafted, their lengths usually shorter, and data compilation more straight forward thus permitting more espedient metanalysis. Then data-driven decisions could be made more readily perhaps garnering the support of NIH despite the lack of robust, full-blown, randomized controlled trials NIH and others seem to prefer (during a pandemic no less).
Why is the NIH voting against Ivermectin then as a safe potentially effective drug against Co-vid? In this day and age where every institution- including medicine- is now plagued with political persuasion, I myself, along with many of my patients, feel there is something rotten in Denmark…AND the NIH for taking such a stance against a drug that is safe, cheap, and with a real potential to do good for patients early in their illness with Co-vid 19 (and now it’s VOC).
Thank you for this. I have been waiting for someone respectable to address this- the silence on this from the medical community only adds to the conspiracy theorists.
I have not prescribed this but have been wondering what the appropriate action is if a patient asked me for this. Given the relatively low side effect profile it seems reasonable to allow them the right to try, especially since I don’t have access to an RCT and I’m not sure any high quality RCTs are underway.
Here is an up t. date (1/4/21) list and analysis of 44 Ivermectin for Covid studies, 17 studies are peer reviewed.
Summary of results
Early treatment ↓87%
44 ivermectin studies
17 peer reviewed
“Early and prophylactic use show high efficacy
100% of studies report positive effects. 87% improvement from meta analysis, p=0.00052.
All studies ↓78%
100% of studies report positive effects.”
Website: https://c19ivermectin.com/
They also have up to date studies of Vit D, Zinc, HCQ, REGN-Cov 2,LY-COV 2. and Redensivir
Sadly, I believe that the reason that there aren’t more trials of ivermectin and other repurposed meds in this country, is that there isn’t money in it, It is terribly unfortunate, but I was unsuccessful in getting an ivermectin trial started early in the pandemic and although no one would come out and say it directly, local institutions were all waiting to participate in “novel” small molecule trials. It just appears that incentives are aligned against repurposing trials.
Risk of Ivermectin is low, and adverse effects pale in comparison to the possible outcomes with COVID-19. I think it is reasonable to try it, and reasonable for prophylaxis in some at risk populations. I, like Dr. Swanson, would not hesitate to use it personally.
I basically agree with Dr. Sax’s conclusions. Actually, I am coordinating one of the rare double blind (Phase 2) trials on ivermectin (https://clinicaltrials.gov/ct2/show/NCT04438850). The study has three arms, placebo and ivermectin at 600 or 1200 µg/Kg, once daily for 5 days. The primary endpoints are safety and reduction of the viral load at Day 7.
Based on Caly’s et al.’s results, we sincerely believe that lower dosages are absolutely unlikely to reduce the viral load in vivo. This is why I admit to being perplexed by the excellent results showed by both observational studies and randomized trials using much lower dosages, often the same ones we currently use for helminthes such as Strongyloides stercoralis. Such regimens, if they really work, should do so for reasons other than reducing viral load. An anti-inflammatory actions has been claimed, which obviously cannot be excluded, however no anti-inflammatory drug has proven successful so far, with the exception of corticosteroids for severe pneumonia and possibly baricitinib.
Unfortunately, obtaining funding for ivermectin trials proved very difficult. Our (unsponsored, spontaneous) trial is proceding slowly (we have now recruited 40 patients while we need 60 for the interim analysis), another reason is the low acceptance rate of patients who may be entirely asymptomatic or have a mild disease not requiring hospitalization (for the 40 who have given their informed consent, we have more than 300 in the subject screening log).
We hope to be able to provide the results of the interim analysis by the end of April at latest (hopefully earlier).
Glad to see discussion of ivermectin for early treatment of COVID-19. Perhaps after consideration of this promising repurposed drug you will reconsider the India Protocol for HCQ PrEP now that Trump is gone and vaccine mania is colliding with reality. Still sad to see so many intelligent professionals who cannot distinguish PrEP from treatment of active disease/infection/PEP. https://www.webwire.com/ViewPressRel.asp?aId=265075
Dear Paul,
Thank you for this level-headed analysis.
I wouldn”t say zero on a study level considering treatmend of Covid in a primary care setting: there’s the (very small) fluvoxamine study and then there’s acetylsalicylic acid.
But nothing at all is approved (and rightly so) in the country were I practice.
This is very unsatisfactory for the practicing GP – What can be given under the ‘not more harm done” policy’? I don’t know. Current treatment regimes in the outpatient setting cover BSC until the patient deteriorates and then they become inpatient.
What would I choose? Maybe ASS? Low dose heparin? I am not sure.
What did I choose until now: Nothing
(Zinc and Vitamin D belong to the supplement category imho)
What we need is vaccination of the populace. What we need are better/bigger trials especially for the outpatient setting.
Other than that I totally concur with the post of Andrew T Pavia.
Why voting against Ivermecrin, while voting with these very costly drugs? We are here in what you described as developing or low income countries, we use ivermectin since start of the pandemic for outpatients, and, it’s doing well. A $1 drug is doing, same as, and may be better than those $100s drugs we are pushed to use, based on what you called” good made studies” . I vote with ivermectin use.
Every article I have seen citing pharmacokinetic considerations for recommending against ivermectin assumes importin a/b1 pathway cited in Caly et al. is the only possible mechanism. That is an unproven assumption that physicians should treat as such. There are other more likely mechanisms that didn’t show up in Caly study due to differences in ACE2 of African green monkey vs. humans. For example: https://pubmed.ncbi.nlm.nih.gov/32871846/ This assumption of Imp a/b1 as only pathway is misguided due to the lack of scientific proof.
Thank you Dr Sax for ypur analysis.
As an ID physician in Latinoamerica ,and trained in USA, Im use to use evidence based-medicine in my practice.
really I think isirrelevant if the drug is cheap, have a low toxicity profile , and so why bnot, why we cannot try.
If we use this empiric approach as scientist ,” the lets try approach bacause is cheap and there´s nothing else collided prom the evidence bsed- medicine that we should use in our practice, or we will begun to use this same principles in another clinicl situations (por example:lets do head CT in all migraines, , its widely available, its relatively cheap ,and may be we can catch some brain tumors or stroke)
Thank you Dr Sax for your analysis.
As an ID physician in Latinoamerica ,and trained in USA, Im use to use evidence based-medicine in my practice.
Really, I think is irrelevant if the drug is cheap, have a low toxicity profile , and so why not, why we cannot try.
If we use this empiric approach as scientist ,” the lets try approach bacause is cheap and there´s nothing else, collided from the evidence based- medicine that we should use in our practice, or we will begun to use this same principles in another clinical situations (por example:lets do head CT in all migraines, , its widely available, its relatively cheap ,and may be we can catch some brain tumors or stroke)
Which HCQ studies are you referring to that say it doesnt work. There are hundreds that say it does indeed work with lots of professionals calling out the studies that they claim is proof that it doesnt work. Ivermecin is much more promising. The problem is the powers to be want the vaccine and it would lose emergency authorization if there are suitable treatments! Prove me wrong lol
Dear Dr. Sax
Nice review and I agree with many of your points. Humanity in its search for an answer to this pandemic that affects us, has tried since the beginning of this disease, to find an answer, unfortunately until now we are, as at the beginning, at zero. my respects for all those responsible professionals who do not forget the scientific method, unfortunately many are carried away by the hope of being the first to have the cure and forget about; do no harm..
Dear Dr. Sax,
Here in Brazil there is a profylatic use of ivermectin also, that have been raising in last months as “probably it does not harm”.
Thank you,
Taciana
Taking a step back, we have to acknowledge that psychology plays a role.
Few expert want to believe of themselves that they’re not using a useful tool. This creates a subtle bias towards discounting potential adoption of a new tool or method.
Few experts want to think that their snap judgements (Spidey-sense, System 1, intuition) can be clouded by emotions. But this happens everywhere else.
That ivermectin feels like HCQ– long videos, laypeople asking for it— or that ivermectin feels odd— how could a pet/ scabies product possibly be applicable?— shouldn’t but does bias the ability to look at the studies. I’ve even seen people suggest that perhaps it only worked if the people had parasites: that definitely looks like an anchoring bias at work [not to mention how does that apply to the Argentina, Egypt, France and Florida studies: parasite burdens can be looked up. They didn’t look.]
Model yourself reading the ivermectin studies as if you didn’t know about the videos, or the veterinarian uses, and especially as if there are no recommendations for or against it. How would a set of neutral medical observers— ones who would feel no personal stress in either direction— review these studies?
Sure, the proponents seem a bit argumentative. So was Barry James Marshall. We cannot forget one lesson of Semmelweis, which is that people strongly dislike the feeling of regret.
Uttar Pradesh has a population of over 200 million (about a sixth of India’s population). They stopped using hydroxychloroquine and started using Ivermectin in September. Have a look at the graphs here:
https://bing.com/covid/local/uttarpradesh_india?vert=graph
Switch the upper right graph to Fatal Cases and the lower graph to Active Cases.
They started pushing out prophylactic kits with Ivermectin, Zinc and Doxycycline to at risk workers (not just Healthcare workers), and later started treating everyone from the household that an infected patient came from. I’d say they’ve done pretty well with their early treatment plan.
https://indianexpress.com/article/opinion/columns/coronavirus-pandemic-covid-vaccine-tracker-uttar-pradesh-7107756/
Ivermectin is being used in at least one hospital in Minneapolis Metro. I have a friend who likely would have died without it. Only one of his lungs was fully functional he found out during his week on a vent. He is a jazz sax player/teacher.
I would ask for it were I infected at any stage. There are plenty studies on youtube. They are from all over the globe. An Indian doctor who is on youtube regularly cites studies from medical publications. He calls himself Dr. Been.
Dr Sax,
Thanks for bringing this potential treatment to our attention!
I have not seen in this discussion thread any mention of the epidemiologic evidence. While is does not prove causality, it does lend added evidence supporting the efficacy of ivermectin for covid-19.
An example from Africa here (https://doi-org.ezp3.lib.umn.edu/10.1016/j.ijantimicag.2020.106248) and another summary from the FLCCC here (https://covid19criticalcare.com/i-mask-prophylaxis-treatment-protocol/epidemiologic-analyses-on-covid19-and-ivermectin/).
We have several months before vaccine use will be widespread, and probably several months before additional RTC evidence is available, particularly if we insist on peer-reviewed publications to guide clinical decision making.
Given that ivermectin is a seemingly safe, low cost, accessible intervention, it makes sense to me to use it now, particularly in the outpatient setting where there are few alternatives.
Some trials compare ivermectin vs HQC, of course you will find differences
Please consider approaching Edenbridge (manufacturer of generic ivermectin) to below the funding of your double blind study. srichardson@edenbridgepharma.com
Ivermectin is widely used in Peru. We had a long first wave and are experiencing a second wave that is coming stronger. There has been a change in ivermectin use since the peak of our first wave in mid July. Then 50-60% of moderate-severe hospitalized patients reported ivermectin use. Now is close to a 100%.
Our ICU’s are collapsing again
Any comment about the meta analysis quoted by Dr. Sax? Strikes me the lack of critical approach. Most of ivermectin trials show astonishing data of improvement compared with controls that go awfully bad. Cero comments about that. Most studies get numbers down by 50-80%… in a disease with quite low mortality and very high spontaneus recovery rates. No critical approach on that. Just good savage comments to fight with Boston colleagues.
Observational experience? A place close to my city Lima is called Ica. Ivermectin has been used there like water for the last 4-5 months. Everybody takes it. We have had the worse excess mortality in the world and Ica is the worst place in Peru. I think part of the culprits of that excess mortality are the advocates of ivermectin and should be occountable for that.
Dr. Gianella,
Thank you for sharing your experience. I have been following the ivermectin debate with some interest and admit I have had a bias towards it because of its association with Paul Marik and his hubris around Vit C, Thiamine and its efficacy. I keep telling people show me the RCT, because I have already been burned with HCQ, plasma, and maybe soon we will be realizing the uselessness of remdesivir as well.
I had theorized that perhaps in some of these studies the parasitic burden in the underlying population might account for differences in outcomes with ivermectin, especially once empiric steroids were used but it is interesting to hear your perspective.
There are many potential treatments of repurposed old drugs and they all have their cheerleaders: HCQ, colchicine, fluvoxamine, now a comment here brought up digoxin. I keep tabs on things to try and give my patients an edge, but I’ve already been burned enough. Most patients, even hospitalized, recover, I’m not going to add to the guilt I already have seeing people die by giving them something that is unproven and later shown to be harmful.
Thanks again,
JP Maxwell, MD
Yakima, WA
So sorry to hear this as the data from Peru did show that Lima was the province with most cases and deaths. However the research and accompanying information suggested that in Lima the drug wasn’t part of the province’s protocol.
Hi Doctor Gianella. Any thoughts on this paper? It seems to contradict your observations. Thank You.
https://www.researchgate.net/publication/344469305_Real-World_Evidence_The_Case_of_Peru_Causality_between_Ivermectin_and_COVID-19_Infection_Fatality_Rate
As a lay person who is a science geek and very proactive with my own health, I find it criminal that a drug with very low risk and potentially high value is not only NOT being recommended by the NIH, but is not being given to every patient in every hospital and every outpatient positive case. People are DYING in droves. What would the harm be in just giving it to them? That they have a side effect? That they die? They are already dying and struggling with all kinds of organ damage. At this point in this pandemic, I am seriously frustrated that the kitchen sink and then some is not being given to people – if it may help and not hurt. Ivermectin, top of the list!
Deborah, I share your extreme frustration because this is definitely a “can’t hurt might help” situation. But some Pharmaceutical company will soon come up with a very expensive version and the companies lobby will sway the FDA.
I have been a member on an FDA panel and remember the “rush to the phones” made by the reporters in the room as soon as we reached any decision.
https://www.bmj.com/content/368/bmj.m1252/rr-6
janet.kelly@northlincs.gov.uk
Early in the pandemic a nursing home reported that all its residents were given ivermectin for an outbreak of scabies. No resident got Covid but staff who hadn’t had the drug did. Since then I’ve followed with growing frustration and anger the lack of urgency in any western country to find a way to stop people from becoming ill enough to be admitted to hospital. In countries with less well developed health care systems it was an imperative and so ivermectin began being used I think starting with Bangladesh. I watched as numbers in those countries began dropping how Africa did not succumb to any great extent and how these countries were bringing more and more (mainly observational) research to the table. It seemed to me to be a no-brained or as we say if it walks like a duck, quacks like a duck it probably is a duck
Doctor Sax,
Thank you for a very reasonable point of view. Here in Brazil, ivermectin and other medications have been promoted (even by our president) as a “silver bullet” against COVID, often with an almost religious zeal. It’s incredibly sad to see the “fake news” mentality applied to science, specially in the middle of such a huge crisis.
I would like to reinforce the idea of “Primum non nocere” – first, do no harm. Between using a medication with possible adverse effects and no benefit in those with COVID, or abstaining from using a medication that may someday be proved to be somewhat helpful, I would always pick the second option.
Dr. Sax,
Thanks for discussing ivermectin. I was surprised that almost no one brought up the fact that ivermectin and HCQ both have synergistic actions with zinc. The negative (often not as negative as purported) HCQ trials mostly used high doses too late, and none that I know of analyzed zinc levels and use. The same may be true for ivermectin. Zinc must be included in any analysis of either drug for COVID. Cheaper early use interventions like vitamin D, ASA, HCQ, and ivermectin need to be used in large trials to adequately measure their effect since a high percentage of early identified infections have good outcomes anyway.
Scott
Dr Hill discusses Ahmed’s paper as evidence that ivermectin shortens viral shedding significantly. This study was conducted in hospitalized patients with mild-moderate disease and no risk factors; patients were randomized to 3 treatment groups (IVM 12 mg/d x 5 days; IVM 12 mg single dose + Azithro 400 mg on day 1, followed by Azithro 200 mg x 4 d; Pbo pills x 5 d) and had nasopharyngeal PCR tests on day 0, 3, 7 and 14. Interestingly, there was no difference in clinical evolution between the two drug treatment groups and placebo group. Aside from the questionnable statistical analysis of viral shedding (mean duration, hazard ratio), what I find most surprising is that 14/23 (61%) patients in the placebo group tested positive at day 14. This is perhaps due to the small sample size, but is inconsistent with the natural course of viral shedding in mild-moderate disease.
Andrew Hill received funding from the WHO to perform a meta-analysis of RCT’s of Ivermectin. He reports unequivocally positive results from 11 studies. https://www.youtube.com/embed/yOAh7GtvcOs
As a practicing FM resident I am frustrated with the amount of patience in some of the posts. Have you all forgotten how successful are anti-viral therapy for other respiratory viral illnesses are currently? I will answer that for you…they are not tremendously successful especially for pt oriented outcomes. Go ahead, tell your patients to use whatever they wish. As for me, I will await for strong evidence to be obtained before I start recommending any snake oils to my patients.