An ongoing dialogue on HIV/AIDS, infectious diseases,
February 3rd, 2019
An “Interview” with the OVIVA Study of Oral vs. IV Antibiotics for Osteomyelitis
An “interview” inspired by publication of a landmark clinical trial. All responses written by me — but be assured, they are based on reading the paper, the accompanying editorial, the supplemental appendix, hundreds of comments on Twitter (some of them from the study investigators), and even a few generous comments from the the senior author in response to email queries.
Me: Thank you for joining us today, and welcome to the official world of published medical literature. We’ve been waiting quite a while with eager anticipation. Can you start out by introducing yourself?
OVIVA: Sure, thanks for having me. My name is OVIVA, which stands for “Oral vs. Intravenous Antibiotics” for bone and joint infections. Clever, eh?
Me: Yes, pretty good — though these makers of this “maple water” might object. Tell me a little about yourself.
OVIVA: Well, as you know, it’s a longstanding view that adults with bone and joint infections need prolonged IV therapy for optimal treatment. But this is inconvenient, expensive, and doesn’t make a whole lot of sense since many antibiotics are well-absorbed when taken orally. Plus, the pediatricians have treated osteomyelitis with oral therapy successfully for years. So we set out to challenge this assumption that you need to use IV.
Me: Great idea! Where was it done, and who did it include?
OVIVA: We did it at 26 sites in Great Britain, from 2010 to 2015 — solidly pre-Brexit — and enrolled around 1000 people. Eligible participants had a bone or joint infection, and would normally be treated with at least 6 weeks of intravenous antibiotics. We included people who had surgery (such as removal of an infected joint or hardware, or debridement), and those who didn’t. The treating physicians chose the antibiotics based on cultures and their clinical practice. These are deliberately very broad inclusion criteria — we wanted to make it as representative of the “real world” as possible.
Me: Interesting choice — really drives home the “pragmatic strategy” in clinical trials design. Any notable exclusions?
OVIVA: We excluded people with Staph aureus bacteremia or endocarditis, since at that time, oral therapy would not have been standard of care — this was way before POET. And since we had to consider the possibility that half the participants would get IV therapy, we did not want to include anyone for whom this treatment might not be completed — hence few (if any) participants were enrolled with active injection drug use. [It’s not clear if anyone who uses injection drugs is in the study.]
Me: So what did you find?
OVIVA: Here’s the big news: Oral therapy was clearly non-inferior to IV. Definitive treatment failure at 1 year occurred in 13% of the oral group and 15% of the IV group. These results held up in several sensitivity analyses, including something we called a “worst case” outcome. In this approach, we take the participants with missing data, and assume all these participants who were randomly assigned to receive oral therapy and no participants who were randomly assigned to receive intravenous therapy had definitive treatment failure. This introduced the worst possible bias against the oral strategy — and it was still non-inferior. Here, take a look at this figure:
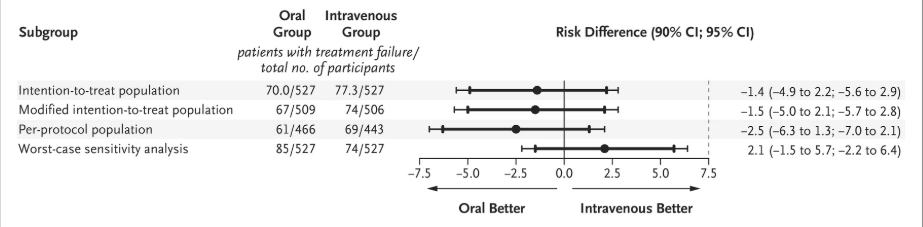
Me: Very reassuring that the point estimates for your baseline analyses all numerically favored oral therapy. And clever job with that “worst case” scenario — good one for the doubters.
OVIVA: Thanks. I should mention that we were worried that the oral group would receive much longer total treatment, but this wasn’t the case — the median duration of therapy was 78 days in the intravenous group and 71 days in the oral group. Plus, even though there was more adjunctive rifampin use in the oral group, outcomes didn’t vary significantly whether this strategy was chosen.
Me: Any other results you’d like to highlight?
OVIVA: Another plus — those getting oral therapy had shorter hospitalizations and fewer complications (in particular, line-related complications). This approach to treatment of osteomyelitis will save plenty of pounds/euros/dollars/etc. So oral therapy can save money, be safer, and be just as effective. Is that what you Yanks would call a home run? Or a touchdown?
Me: Vastly prefer the former, thank you. And no doubt these are great results — congratulations for being such a challenging, important, and rigorously conducted study. Highly likely to change clinical practice!
OVIVA (beaming): Aw shucks — that’s very kind of you.
Me: However, as as noted in this excellent editorial, it might take a while — this particular study might not be enough for certain clinicians, and earlier studies were smaller (or forgotten). And some of us might have trouble convincing our surgical colleagues that their patient with osteomyelitis can be treated with “only” oral antibiotics after debridement — for many surgeons, “more” equals “better.”
OVIVA: Yes, our surgeons are like your surgeons.
Me: May I raise a few additional questions and concerns?
OVIVA: Of course — this is your blog, and you’re writing this made-up interview!
Me: I noted that the oral regimens selected by the clinicians were mostly quinolones, doxycycline, and clindamycin — relatively few got oral penicillins, and oral cephalosporins didn’t get chosen at all. And no love for trimethoprim-sulfamethoxazole in Britain? No linezolid?
OVIVA: We did not mandate antibiotic selection — the antibiotics chosen represented clinical practice at the sites. Oral cephalosporins are hardly ever used in Britain, and not enough people chose “cotrimoxazole” (that’s what we call it) to warrant a separate line-item in the report. Some chose linezolid, but not for more than two weeks.
Me: Understood. But here’s another way of looking at the results — if clinicians generally select highly bioavailable oral antibiotics for treatment of bone and joint infections, then oral is non-inferior to IV. This might be overinterpreting the subgroup analysis, but the forest plot for planned oral treatment even hints at this conclusion:
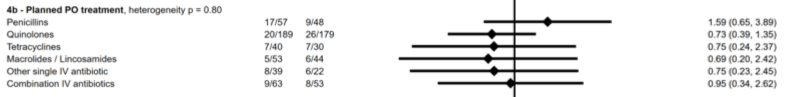
OVIVA: Hey, you read the Supplemental Appendix! Very impressed. In my defense, let me quote from the paper, which states: “We did not seek to compare specific antibiotic agents or to stipulate which agents should be used.” Someone else might choose to do that sort of study. We simply can’t say whether one oral strategy was better than another.
Me: Got it. But clinicians will read this paper and want guidance over what specific antibiotic regimen to use for a specific bug for a specific indication — this study can only give the broad view that oral is non-inferior to IV. Not much granularity here. And from a practical standard, we’d all appreciate more information about dosing.
OVIVA: Hey, I can’t be all things to all people. Rest assured, more analyses and reports are coming. And remember, we relied on the expertise of the ID docs at the sites, whom I assume were among the smartest — if not the smartest — clinicians responsible for choosing the treatment.
Me: No argument from me on that point.


All my love to trimethoprim-sulfamethoxazole….
Thanks for this, Paul. This will be good news for my patients with diabetes who develop a foot infection and then osteomyelitis. Of course, that is assuming using oral antibiotics for osteomyelitis becomes adopted as a practice…. Disappointing that both POET and OVIVA enrolled few, if any, IV drug users. That’s pretty “real world” here in the US. Maybe not in the UK? Also disappointing that certain antibiotic drug classes were not represented in the study, but heck, a study can’t be all things to all people, right? I’m just glad we may get away from exclusive use of IV antibiotics for osteomyelitis at some point.
Hi Paul, I saw your blog on the OVIVA trial, it was brilliant!
Do you know why it took so long to publish? I went to the MSIS meeting in Boston in March of 2017 where the PI presented pretty much what the trial showed, and was super excited and waited, and waited and waited, still no OVIVA…..
I was starting to conclude that it must have serious flaws when this NEJM came out with both that and the POET trial in the same issue. In my opinion the two most important ID studies in the last couple of years at the same time.
Thanks, Roger. Per the senior author, there was a lot of back-and-forth on the recommended revisions.
Good to see it finally published!
Paul
HI paul – great review. 2 questions 1) was trying to tease out from the data average time on Iv therapy prior to switch..IRL we usually start IV until we know sensitivities (if we have them). But was there a group that got empirically started on oral? or was everyone on IV at the beginning 2)Can you comment more on the use of rifampin with oral therapy . from what i can tell it didnt make a big difference? hard to tell…
Hi Lisa, thanks —
1) Looks like they were allowed 5 days of IV before the randomization.
2) Rifampin was used based on clinician preference. It didn’t make a difference in this study, but it wasn’t a randomized trial of rif vs no rif, so not definitive one way or another!
Paul
Oral therapy following IV treatment for patients with osteomyelitis from contiguous spread of infection: Amoxicillin-clavulanate 875 mg/125 mg or Ciprofloxacin 750 mg plus clindamycin 300-450 mg or Levofloxacin 750 mg PO daily plus clindamycin 300-450 mg…….(mygenericpharmacy) Deepak Sharma
I balk at the idea of using clindamycin for 6 weeks . Is it something most ID physicians use for 6 weeks without compunctions ??
Well, I have indeed eagerly awaited this trial, but always concerned it was a bit like an RCT of parachutes. Given the antimicrobial activities and pharmacokinetics of available oral agents, did anyone suspect a different outcome? Just askin’.
Cheers,
Old ID guy who has used a lot of PO meds for this indication over the decades.
Nonclinical post here: this is entertainingly similar to the NPR ‘Everything is Alive’ podcast, which I think you should request to be a guest speaker on, perhaps representing some infection or antibiotic, possibly vaccine?