An ongoing dialogue on HIV/AIDS, infectious diseases,
April 12th, 2022
Should We Prescribe Nirmatrelvir/r (Paxlovid) to Low-Risk COVID-19 Patients?

I usually put a clever caption below these vintage public health posters, but can’t think of one. Happy to take suggestions!
The top recommended treatment for high-risk outpatients with COVID-19 in the NIH Guidelines is nirmatrelvir/r (Paxlovid). It’s quite clear why.
In the EPIC-HR study, unvaccinated people at high risk for severe outcomes had an 89% reduction in the risk for hospitalization or death compared to placebo. If we just look at mortality — another important endpoint, don’t you think? — nirmatrelvir/r beat out placebo by a score of 0 (nirmatrelvir/r) to 13 (placebo).
Let’s add to this very favorable outcome several other benefits:
- The reliable activity against all variants
- The relatively good safety profile
- The short course of treatment (5 days)
- The substantial reduction in viral load, the most of any drug tested to date
- That it is pills rather than an IV
Yes, folks, we have a winner! Sure, it has lots of drug interactions and it tastes terrible, but it’s far and away the best choice out there right now.
So if we recommend this treatment for high-risk people with COVID, what about for symptomatic people who are not at high risk? Not now, but imagine a time when we had sufficient supply. Should we also recommend it for them?
I thought the answer was straightforward, and will give my views below. But I had an inkling that this wasn’t so clear a few weeks ago when one of my smart colleagues held the opposite opinion.
To test these choppy waters, I posted this poll online:
Let's suppose there's an ample supply of nirmatrelvir/r (Paxlovid), and you can prescribe it for anyone with symptomatic COVID-19. Cost not an issue. Based on what we know today, would you do so for "low-risk" cases? Why or why not?
— Paul Sax (@PaulSaxMD) April 10, 2022
Wow. Not only is there a pretty even split, but responses are so interesting, and quite strongly held. It’s fascinating to read them.
(And, by the way Dr. Titanji, good call.)
My own view? Given sufficient supply — and we’re not there yet — I’d certainly recommend nirmatrelvir/r even for low-risk symptomatic people. The motivation lies in the already summarized favorable results of the high-risk study, and hinted at even in the interim analysis of EPIC-SR, the study in low-risk people.
Remember, some low-risk younger people get severe disease. (Here’s a notable recent example — speedy recovery!) The reasons are poorly understood why this happens to some unlucky few (OK, not understood at all), and it’s rare, but all of us have seen these unfortunate cases. Could nirmatrelvir/r reduce the risk for severe disease even in this population?
Highly plausible. Look at the interim results of EPIC-SR which, though not showing benefit in the primary endpoint of time to symptom resolution, did appear to yield clinical benefits for this prespecified clinical endpoint:
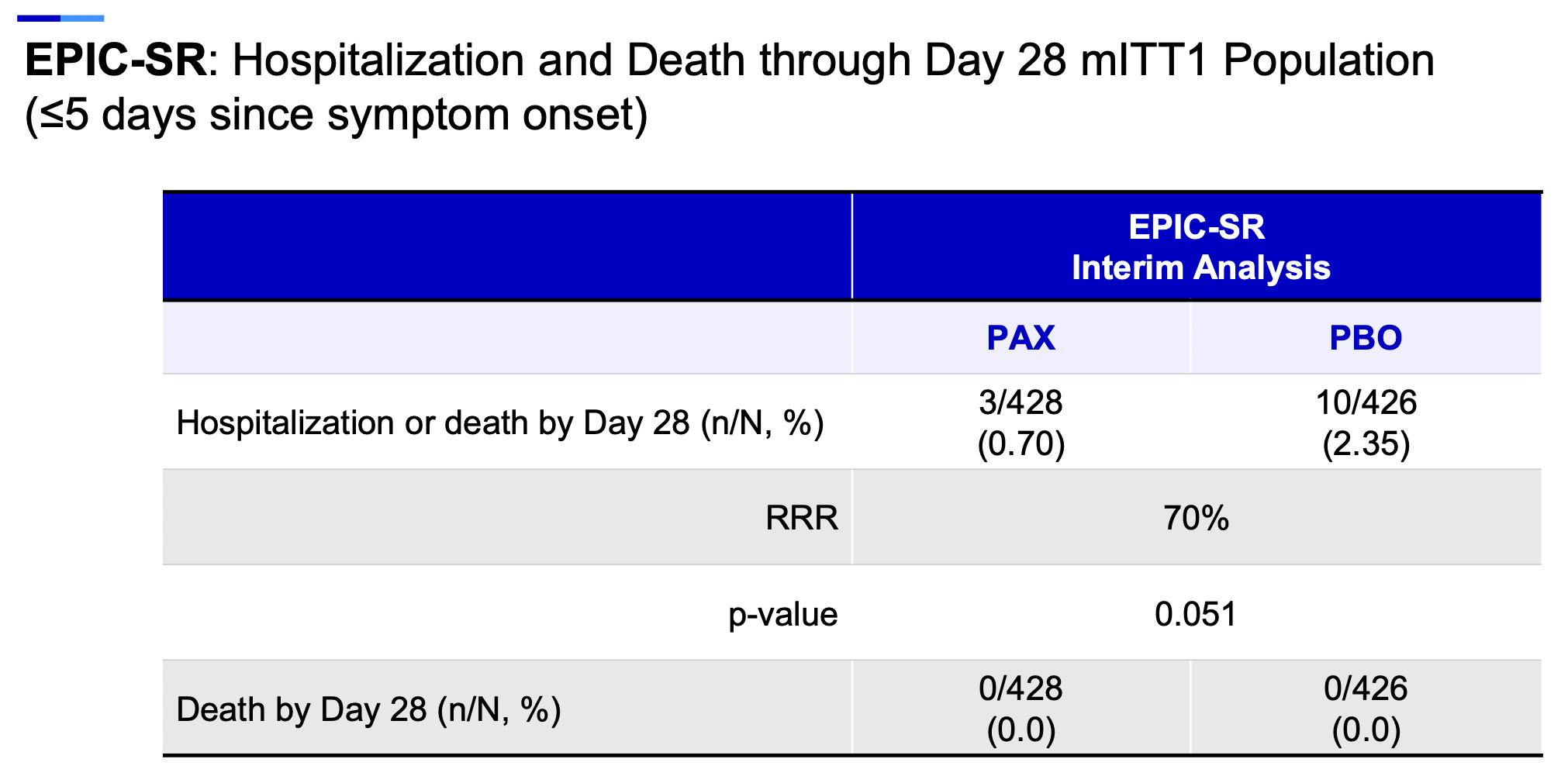
A further advantage is the virologic response, which should reduce the likelihood of onward transmission. For those needing negative antigen tests prior to returning to work or school, it’s another plus to hasten this process.
Also, there’s the theoretical benefit that treatment will reduce the risk for long COVID, or other prolonged post-infectious symptoms. Both most certainly occur in people at low-risk for severe disease. Since at its genesis these are virus-induced complications, it’s not crazy to think that inhibiting and shortening viral replication will make these dreaded outcomes less likely. It will be challenging (and take some time) to prove, but it’s an important part of the research agenda.
Finally, treating symptomatic COVID-19 falls right in line with the first principles of our specialty. If there’s a safe, effective, and readily available treatment or prevention strategy for a symptomatic infection that is potentially serious, we treat it — even if most people will do fine.
I’m not alone in my view, obviously — 52% of respondents agreed. One respected ID researcher (who recently experienced her own household outbreak that was “not fun”), emailed me:
I agree with you 100%! I almost commented on that poll to say I thought it was a no-brainer but then saw how many people disagreed and stayed quiet! If it cuts short viral replication, the impact on secondary transmission could be enormous — far more than our vaccine responses are doing. What a huge benefit that could be!
The opposing view says it’s not yet been shown to benefit this population of lower-risk people. That these clinical endpoint results are “fragile” — so few outcomes that it could be an accident (and maybe motivated the increased sample size in the EPIC-SR study). That even if there is a clinical benefit, the number needed to treat will be gigantic. That resistance may develop. That the reduction in transmission effect isn’t proven. That there are places that don’t have this treatment at all, and our indiscriminate prescribing of nirmatrelvir/r will prevent it from being available abroad.
Lots of echoes of the oseltamivir controversies over the years. These are valid concerns, all of them. So I really do get this opposing view.
It’s just not mine.
What do you think?


Paxlovid is an extremely important treatment for immuno-compromised patients. Both for those individuals and for society, since their long infections might be a source of new variants. Since there is little to no evidence that infection fatality rate is correlated with transmissibility in SARS-CoV-2, new variants are likely to be hugely important as the pandemic proceeds. Thus, as an evolutionary biologist, I would want to know more about the relative risk of evolution of resistance to Paxlovid. Firstly, some data suggests that a high proportion of transmission happens before symptoms develop. This might reduce the epidemiological value of treating low-risk patients with Paxlovid. OTOH, this could also reduce the selection pressure for resistance to Paxlovid. Hard to know which will be a bigger factor. Further, there is little data on the benefit (e.g. reduced morbidity, risk of long covid or other long-term sequelae) of treating low-risk patients. Without more information on all these points, it’s currently difficult to know what is best. The clearest point is that more research funds are needed to answer all these questions!
For the poster:
The goal – Beat Covid
The starting blocks – Ventilation, Masking, Vaccination
For the caption:
Should Nirmatrelvir be the fourth lane?
Sorry . . . couldn’t resist . . . .
Very clever, Gillian. Well done.
Thanks Gillian! Next time I’ll ask you before posting. 🙂
-Paul
I am a young triple mRNA vaxxed low risk patient who took Paxlovid. I took a rapid test every day of my infection and after starting the meds (day 3 of symptoms) it took 3 days and I was testing negative again. I felt better very quickly which meant that I could care for my youngest child who also tested positive which allowed my partner to isolate from us with our older child. It prevented them from getting it. I went from very sick to almost 95% normal within 24-36 hours, which has a lot of value in my opinion. I took it to try to avoid long Covid mainly. As long as widespread uses doesn’t lead to resistance I don’t see the issue.
This outcome is the norm with 99.9% of patient with low risk and fully Vaxxed , without any expensive treatment. i think we should keep it for high risk patient , more cost effective .
Can we get more data?
The benefit of of Paxlovid was seen in a high risk unvaccinated population. The same or even higher level of protection against severe illness and death is seen with vaccination.
What is the benefit in a vaccinated population?
Most of my patients are vaccinated and I’m not sure how best to use the supply of Paxlovid I have. I have no data to guide me.
How did you manage to obtain the Paxlovid? Current regulations don’t permit young, low risk patients access to Paxlovid
Excellent review, as usual. Balanced, and with a throrough anaylisis of all potential benefits and drawbacks of giving Nirmatrelvir/r (Paxlovid) to low-risk individuals with early COVID.
I would just add that all these data support carrying out a double-blind RCT to assess if the final trade-off between benefits and risks back its administration in this scenario.
However, they do not support prescribing the drug in these subset of individuals.
Also would like to see the benefit in clinical endpoints, the cost-effectiveness analysis, and the number needed-to-treat to avoid one hospitalization or a severe outcome.
Josep M Llibre
@DrBike7
Hi Paul,
In my opinion, what we lack as physicians and in general as humans, is restraint. Why is this strong urge and compulsion to “do something” all the time? When Penicillin was first discovered, it saved lives but then when used for every little infection, what was the outcome? Let’s have the courage to give the human immune system a chance and leave antivirals for those who badly need them.
In terms of resistance developing with widespread administration- I would think that giving Paxlovid to low risk patients, who are likely to rapidly clear their infectious virus, presents a far lower risk of resistance development than giving it to immunocompromised patients (who obviously should still get it!) Protease inhibitor resistance will likely come with a fitness penalty, at least initially, and seems way more likely to evolve over time in a permissive host whose immune system is unable to clear the infectious virus. My fear is that it will arise in someone with persistent infectious COVID, multiple short Paxlovid courses or one inappropriately long course, maybe with subtherapeutic drug levels from emesis or diarrhea relating to COVID or the medication itself. Fear of developing resistance doesn’t seem like a great reason to withhold this drug from low-risk people.
Great posting, Paul, and excellent comments on both sides. As with virtually every treatment given EUA, there isn’t enough data to answer all the important questions. Performing a RCT for low-risk patients is absolutely necessary, and it will take time to gather the much needed outcomes data on long COVID. Also, given the multiple drug-drug interactions (DDI) with Paxlovid, the NNT to harm one individual from DDI should also be tracked, as that could prove to be a significant competitor to benefit as the NNT to benefit one patient rises with treatment of a lower risk population (typically in those circumstances, NNTB rises while NNTH remains the same).
So here is a question I do not think I have seen addressed (using approximate numbers to clarify the issue): If a drug is 90% effective for an important endpoint when tested in an unvaccinated population, and vaccination itself is also 90% effective for that same endpoint, can we conclude that the drug would reduce that endpoint by another 90% if given to vaccinated people who have a breakthrough infection (or would the drug perhaps offer no benefit to vaccinated persons)? Would love to have data, but even a discussion of this topic would be of interest.
Great post Paul,
i will add the cost of the pill and the lack of evidence on Cons
i will use assess the use in in householder of patients with rick of severe clinical progression
PS all evidence come from RCT of largely unvaccinated patients. It’s time to move forward other populations, otherwise we probably use a new drug for no more existing patients
Hi Paul, thanks for this review.
The question that has been puzzling me lately is whether this therapy should be offered to symptomatic VACCINATED individuals, especially those in the high risk category. It seems like EPIC has focused on unvaccinated individuals (which may have unintended side effect of having these individuals justify NOT getting vaccinated if this highly efficacious therapy is available), but are there any plans to study this in vaccinated folks? Or can we just use these results to extrapolate? Given the choice, would you rather treat an unvaccinated low-risk young adult versus a vaccinated high-risk/older adult?
Covid is opportunistic, and seems to find unknown co-morbidities. You never know what is ticking inside you, and we keep learning of new effects of Covid, and it seems to spread throughout the body with a large percentage of the people it infects.
This is a non-covid example, but a girl I knew in college became an Olympic distance runner medalist. At 50, she had a pulse rate of 28, and had a double pulmonary embolism. She survived, fortunately, but she appeared to be healthier than anyone. But something was ticking inside her.
I just read of a thin, supposedly healthy 39-year-old who caught Covid and now needs a heart transplant (he was unvaccinated).
The side effect profile of It Paxlovid makes it appear quite safe. The safety profile should be the decision-maker, even with efficacy level of 50%. This one is north of 80%.
I’m a practicing internist in Southern California. Paxlovid is readily available at many local pharmacies. I am certainly in the camp of prescribing Paxlovid for any symptomatic patient. I am an advocate for my patients. While I strongly believe that we should make this drug easily accessible worldwide-my prescribing this drug locally at this time will have ZERO impact on anybody getting this drug outside of my community at this time. Additionally, my elderly/immune impaired patients with limited transportation and with home test kits-I have given a few prescriptions as to avoid any delay in treatment in the event of getting ill. One of my patients is going to Antarctica for 6 months in the fall and feel it is ethical for her to have Paxlovid in case of an outbreak when she arrives there (with home test kits in tow).
AHP MD
I’m moderate risk (asthma) with a pregnant spouse, both fully vaccinated. I had a negative and then positive PCR within a day of each other and started Paxlovid on day 3 (day after positive PCR) with the goal of reducing transmission risk/presumed reduced viral load. Though still practicing strict isolation and quarantine protocols, masking indoors and will for 10 days regardless of antigen test. We also have the benefit of weather on our side, being able to keep all the windows open and ventilation running. I know you didn’t ask for non medical opinions but sharing a situation where my medical team and I made what I think is a good decision.
Thanks for the write-up Paul. I am a 3x vaccinated, young male with very mild asthma. My doctor prescribed paxlovid for me upon infection and said he would have recommended it even if I didn’t have asthma. I have a couple lingering thoughts around it:
1. Is the expectation that it would be prescribed multiple times in the event of another discrete infection?
2. Does the protease immediately return to normal after it has been blocked by this drug? Or is there risk that it will remain inhibited?
I am a 72 yo Id fit I’d . For all of the reasons above when I tested positive on day two of symptoms after two negative home tests Los pcr . I took paxlovid for 5 days . Day 6 antigen and pcr neg and no symptoms day 8 return of symptoms and positive antigen which remained positive until day 18
Wife 50 with one risk factor had similar experience .all better now but it certainly seems like a rebound and goal of reducing viral load did not happen
Great and relevant discussion. My question for the group is how early or how late should you give it? Is starting it 5 days into symptomatic covid in a vaccinated low risk patients reasonable or should we use the Tamiflu analogy and offer it within the first 48 hrs of symptoms?
Thank you for an interesting read, Paul. I am very hesitant to give Paxlovid to my triple vaxed low risk patients, because the trial that showed such a large benefit occurred in an unvaccinated population. The benefit in triple vaxed patients might be vanishingly small. Perhaps the NNT is low-risk, triple vaxed is >100k? Do we know that this new medication is so safe to justify such a large NNT? To me, this seems like the sort of thing that we physician should be demanding Pfizer pony up to demonstrate in a large RCT. Maybe they could reinvest some of their insane profits to provide us with good information, instead of us just guessing about whether to give this to everybody?
Clearly, the next step is a randomized trial of low risk SARS cov 2 infected using a 5 day or possibly 3 day regimen and looking at
1. clinical response
2. viral shedding parameters
3. reduction in transmission to uninfected contacts
For now, Paxlovid is a good addition to our therapeutic choices.
I expect that physicians using Paxlovid will have a chance to share their experience
Great article
It all sounds well and good to prescribe Paxlovid to everyone. However are we interfering with natural immunity and just like the kid who was never exposed to peanuts and now as an older child is deathly allergic to it . This is where natural immunity can be very helpful and protect us going forward . Especially given the latest itineration. I agree for moderate to high risk patients however for low risk patients I think vaccinations and natural immunity is our best way forward .
I am 67 yo male with no other co-morbidities except (controlled) hypertension. I am 5 days past onset (4 days past +ve antigen test). My question is: is it still beneficial to take paxlovid to avoid (a) possibility of it taking a severe turn, and (b) avoiding risks of long covid.
I am very low risk and didn’t qualify as high risk. My doctor did advise for me to take it.He explain shortening viral replication to possibly reduce the risk of long haulers is what I understood.
I also have a two high risk individuals in my home and I am nurse.
I will say that I have taken care of and know of healthy people with no co-morbidities that have developed long-haulers syndrome.
Taking my last dose tomorrow and have no sxs just the metallic taste in my mouth since the first dose.
Stay well everyone ✌