August 5th, 2013
Putting the 2013 Heart Failure Guidelines Into Practice
Clyde Yancy, MD, Harlan M. Krumholz, MD, SM and John Ryan, MD
CardioExchange’s Harlan Krumholz and John Ryan interview Clyde W. Yancy, lead member of the ACCF/AHA task force that wrote the 2013 guidelines for management of heart failure.
Krumholz and Ryan: What is the biggest challenge to implementing the heart failure guidelines for every patient with heart failure?
Yancy: The biggest challenge is no longer in establishing “proof.” The database we now have is robust, and the work that we and others have done make a compelling argument that guideline adherence works. Thus, the biggest challenge is operational — namely, how to make the information easier to deploy and access. The parent committee’s strategy is the concept of GDMT, or guideline-directed medical therapy. The goal is to use simple algorithms to clarify the best application of evidence-based, guideline-directed therapy for patients with cardiovascular disease, according to appropriate indications.
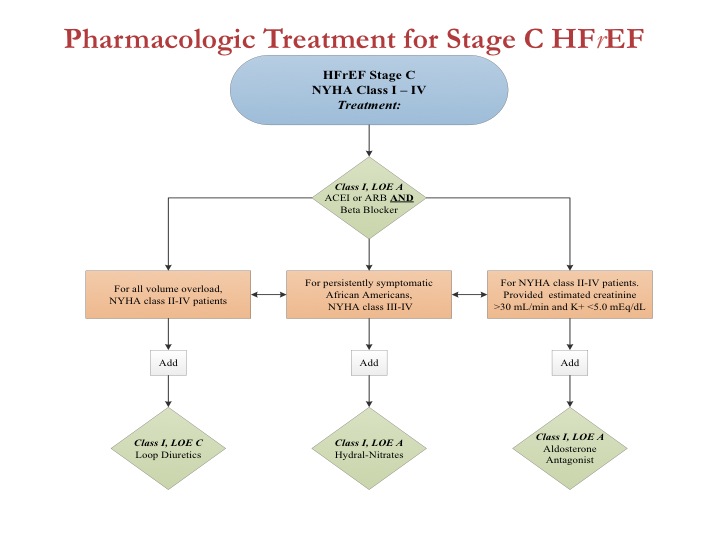
Figure 1 from the heart failure guidelines, shown here, is the linchpin of this effort. In a single graphic, we represent the best application of medical therapies for patients with heart failure and reduced left ventricular ejection fraction (HFrEF).
Click here to download PowerPoint slide
In addition, we have a similar graphic for device therapy and an expanded emphasis on HF with preserved left ventricular EF (HFpEF), including specific applicable guideline statements. If the deployment of GDMT works as intended, we will move closer to broad implementation of best therapies for HF.
Click here to download PowerPoint slide
Click here to download PowerPoint slide
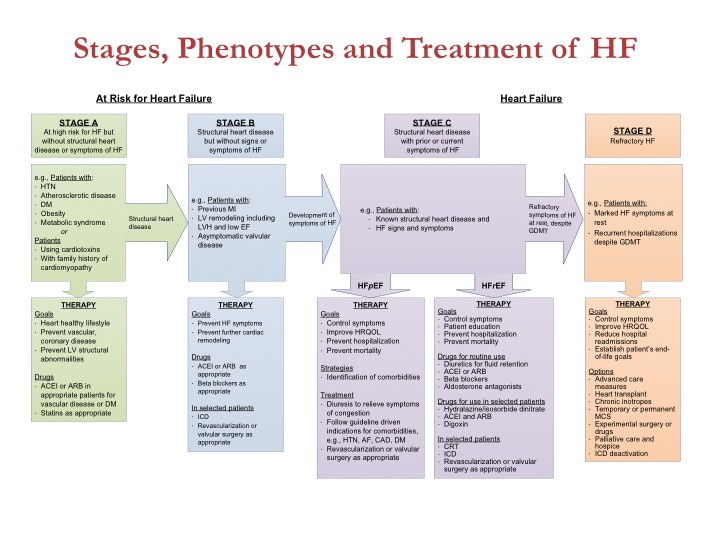
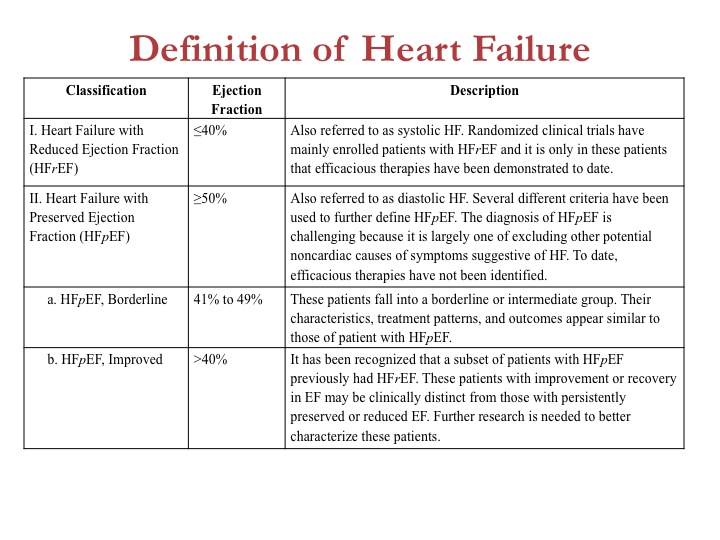
Krumholz and Ryan: Heart failure with preserved LVEF is now defined in categories: HFpEF (EF >50%); HFpEF, borderline (EF 41–49%); and HFpEF, improved (EF >40%). How often should patients undergo an echocardiogram to identify their HFpEF or HFrEF category? And should patients be managed according to their current EF or their worst historical EF?
Yancy: Great questions. We consider it important to identify the increasing number of patients formerly with HFrEF who have recovered sufficiently to now have an EF >40%. This group has not been well studied, but a good guideline document answers some questions and raises others. We hope that by defining these cohorts of HFpEF, we will spur investigators to study each subgroup.
Click here to download PowerPoint slide
Can we stop or reduce GDMT in patients who have recovered their ventricular function? If a device needs a generator change but the EF is now >35%, is the change still necessary? We don’t know the answers to those questions, but without making this distinction we would never pursue the answers. For now, as we say in Texas, “Dance with the horse that brought you to the party.” In other words, we don’t advise switching a regimen that appears to have promoted an improvement in ventricular function. And only check ventricular function when the clinical status has changed or an intervention intended to affect LV performance has been initiated.
Krumholz and Ryan: You acknowledge the increasing cost of treating patients with HF. How do you propose those costs be controlled, especially as use of mechanical circulatory support (LVADs) expands?
Yancy: So this is the tough question. As a guiding principle, the ACCF/AHA guidelines are written without regard to cost. That is less of a copout and more an acknowledgment that our database is surprisingly rudimentary regarding cost-effectiveness. We know more about the cost-effectiveness of LVADs than we do for sodium restriction or parenteral diuretics. But we do have a fairly strong gestalt sense that quality care, as driven by our guidelines, is cost-effective care, given that we put primacy on the preservation of life.
Your question specifically mentions LVADs. At present, they are not cost-effective, but the numbers have dropped from >$800,000 to <$150,000 per QALY. That is still not considered cost-effective, but the progress has been monumental. The more important point, however, is that even with the current use of about 1500 LVADs per year, this is an infinitesimally small fraction of the overall HF cost bucket. If LVADs went away altogether, we would still spend billions of dollars on heart failure. Accounting for what we can do elsewhere with more-appropriate use of hospital resources and true disease prevention, the best application of medical and device therapy will matter much more than the isolated use of LVADs.
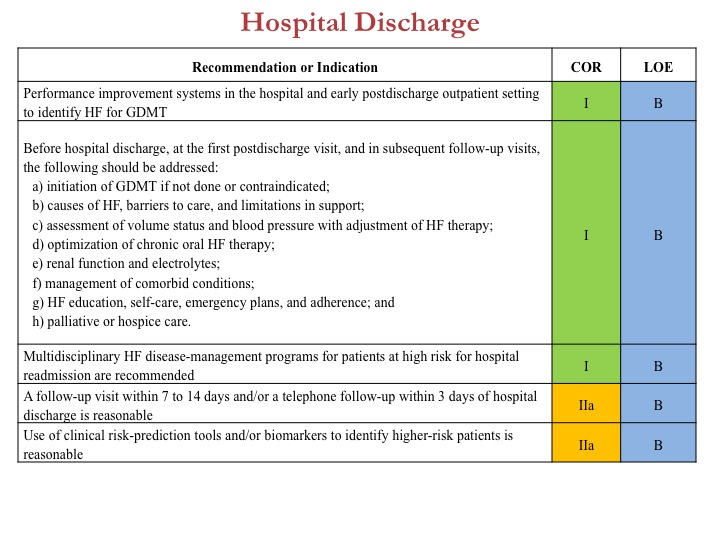
Case in point would be best deployment of evidence-based strategies to reduce readmission after heat failure hospitalizations. Taken together, these new guidelines are intended to present an updated evidence-based approach that will optimize best outcomes in heart failure, identify residual questions in heart failure, and strive for best quality.
Click here to download PowerPoint slide
To review all the content, tables, and figures from the new ACCF/AHA 2013 heart failure guidelines, click here.
JOIN THE DISCUSSION
Share your observations about the new heart failure guidelines and Dr. Yancy’s reflections on them.

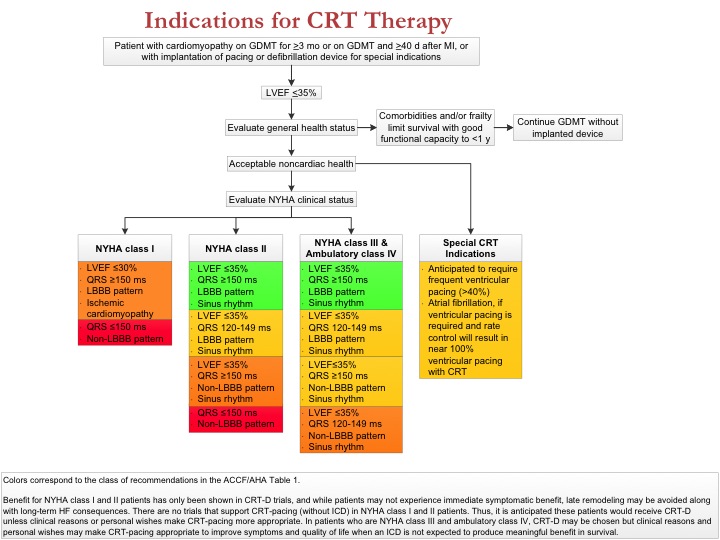
For CRT patients, studies have shown a significant incremental benefit as the percentage of biventricularly paced beats increases throughout the 90-100% range. This suggests a negative impact of native LBBB conduction, event if well less than 10% frequency. Nevertheless, CRT is recommended for HFrEF patients requiring RV pacing and expected to pace at least 40% of the time. Is this a paradox? Is this based on the DAVID trial? Is RV pacing on average less dysynchronous compared with native LBBB conduction?
Great and clear algoritms.
remain about cardiac pacing RCT and implanted cardiovertor.
ICD indication is frequently a difficult situation (In France specially) in very sick and old patients Will it helps the patient?How to ex plain to the patient (and family) that we eventually may swich it off in terminal situation?
We are furthermore never sure that there is not an underlying disease which will impair the longevity below one year. Cancer can emerge also.
However these guidelines help me (us) very much and also for the follow-up always a challengein long term.
Thank you.
PS: one remark one the first algorithm (Pharmacologic treatment) we should read potassium<5 meq/l, not <5 meq/dl.
First, heart failure is the failure of the whole heart. Therefore I think the definition of HF should include LA performance (size and function as well) besides EF because LVEF is linked to LA function. Second, special population are patients with atrial fibrillation – in my experience, cardilogists perform ECHO only after fast ventricular rate had been slowed down and by that time volume overload had been decreased or eliminated. These EF results in compensated HF pts cannot always be helpful then.