September 11th, 2013
Oral Anticoagulation, Part I: Direct Thrombin Inhibitors
Akhil Narang, M.D.
When I started residency 4 years ago, warfarin was really the only choice of anticoagulation widely used for prevention of stroke in patients with atrial fibrillation (AF) and in patients with venous thromboembolism (VTE). Despite knowing about the coagulation cascade for decades, only recently have viable alternatives to warfarin become available. In this post, I hope to break down (at a macroscopic level) use of oral direct thrombin inhibitors (DTI). In my next post, I will review Factor Xa inhibitors.
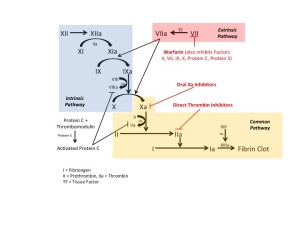
To start, let’s review the basics of clotting (Figure below). Fibrin clots form after activation of the intrinsic and extrinsic pathways. Tissue injury initiates the intrinsic pathway and compromises the vast majority of the coagulation cascade. Warfarin inhibits vitamin-K dependent cofactors (II, VII, IV, X) in addition to protein C and protein S. Warfarin acts across both the extrinsic and intrinsic pathways to prevent thrombus formation. Direct thrombin inhibitors and Factor Xa inhibitors both work in the common pathway of coagulation.
Most DTIs are administered parenterally and used in patients with ACS and in treatment of patients with heparin-induced thrombocytopenia. Oral DTIs have been slower to reach the market. While several are under development, dabigatran received FDA approval in 2010 for prevention of stroke in patients with nonvalvular AF. The RE-LY trial, published in 2009,showed that low-dose dabigatran (110 mg twice daily) was noninferior to warfarin for preventing stroke in patients with intermediate risk nonvalvular AF (mean CHADS2 score 2.1). High-dose dabigatran (150 mg twice daily) was superior to warfarin in this population. The risk for major bleeding (compared with warfarin) was lower in the low-dose dabigatran group and similar in the high-dose dabigatran group. In 2013, the RELY-ABLE study, which extended the follow-up period (median of 4+ years) for patients in the RELY trial, showed similar rates of stroke and death in both groups of dabigatran. In another recent analysis of the RE-LY population, in which cardiovascular endpoints were weighted, researchers concluded that both doses of dabigatran have comparable benefits in prevention of stroke when taking into account safety and efficacy.
The bottom line is that dabigatran is efficacious for stroke prevention in nonvalvular AF patients. Not having to monitor one’s diet or monitor INR are practical advantages that dabigatran offers over warfarin. The inability to readily reverse bleeding associated with dabigatran should be a point of discussion with patients when choosing the appropriate anti-coagulation strategy (especially in elders or patients with histories of bleeding), however a dabigatran antidote will likely be available in the future.
Turning towards venous thromboembolism, dabigatran has been investigated for treatment of acute VTE and also for prevention of recurrent VTE. In both scenarios, dabigatran has shown positive results. In the RE-COVER study, published in 2009, patients with acute VTE were bridged with either warfarin or dabigatran (150 mg twice daily). No significant difference was found in the rates of recurrent VTE or major bleeding. Further trials (RE-MEDY and RE-SONATE) evaluated dabigatran versus warfarin or placebo in patients who completed at least 3 months of treatment for acute VTE. The results showed dabigatran treated patients had similar rates of recurrent VTE (and lower rates of major bleeding) than those on warfarin. Interestingly, a higher incidence of ACS was seen in the dabigatran-treated group. Less surprisingly, when compared with placebo, dabigatran treated patients had a lower rate of recurrent VTE (and higher rates of bleeding). As of now, dabigatran has not been FDA approved for treatment or prophylaxis of acute venous thromboembolism but is being used off-label for these indications by some clinicians.
To date, dabigatran is the sole oral DTI available in the U.S. and, as such, controls the majority market share in this class of medications. I anticipate newer generations of DTIs in the coming years. Stay tuned for the next post that will address Factor Xa inhibitors and their role in the treatment of atrial fibrillation and venous thromboembolism.


Well, in the US majority market share controls Xarelto.
Nice.keep it up